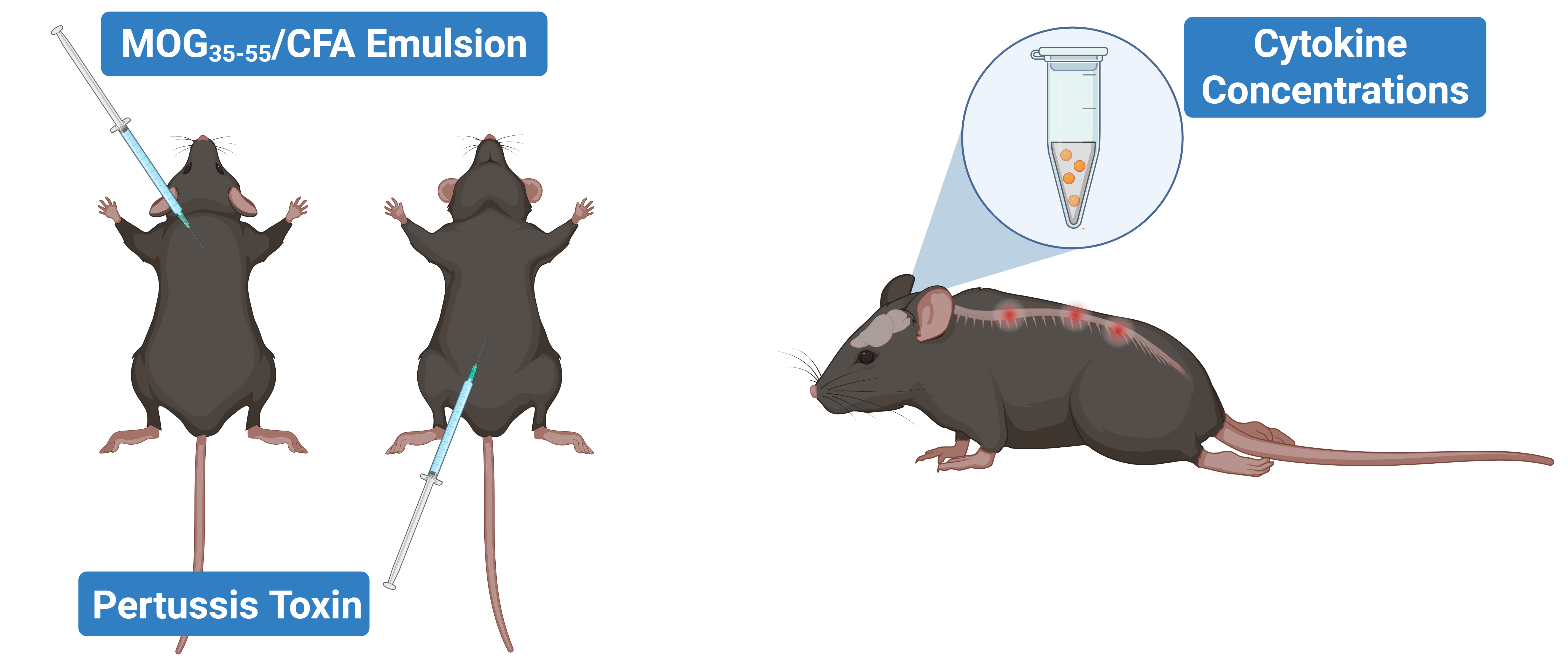
Neuroinflammation in the EAE Mouse Model: mIF & Cytokine Analysis
Table of Contents
Cytokine Analysis & Multiplex IF in EAE – An Interactive Presentation
Research Study Summary: Cytokine Profiling in EAE
We directly measured cytokine protein levels across multiple biological matrices (both CNS tissues and fluids) in EAE to capture an aspect of the immune response that is frequently inferred, but less often quantified at the protein level. We induced EAE in mice using MOG35–55 in complete Freund’s adjuvant (CFA) with pertussis toxin, and disease was assessed 21 days post-immunization. Cytokine levels (IL-1β, TNF-α, IFN-γ, IL-6, IL-10, IL-4, IL-2, IL-5, and the chemokine KC/GRO) were quantified using an ultrahigh sensitivity multiplex immunoassay in plasma, CSF, and spinal cord homogenates.
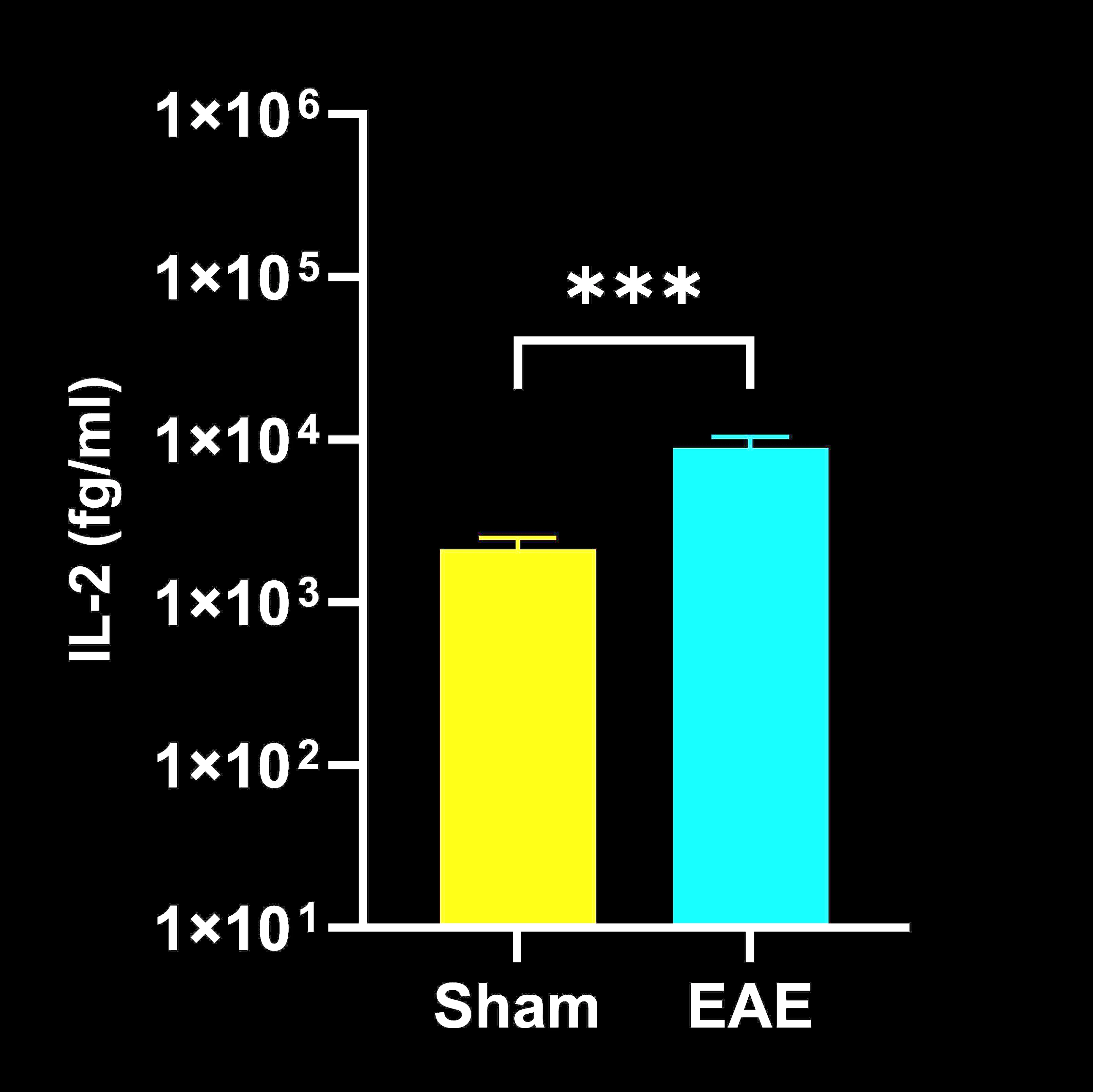
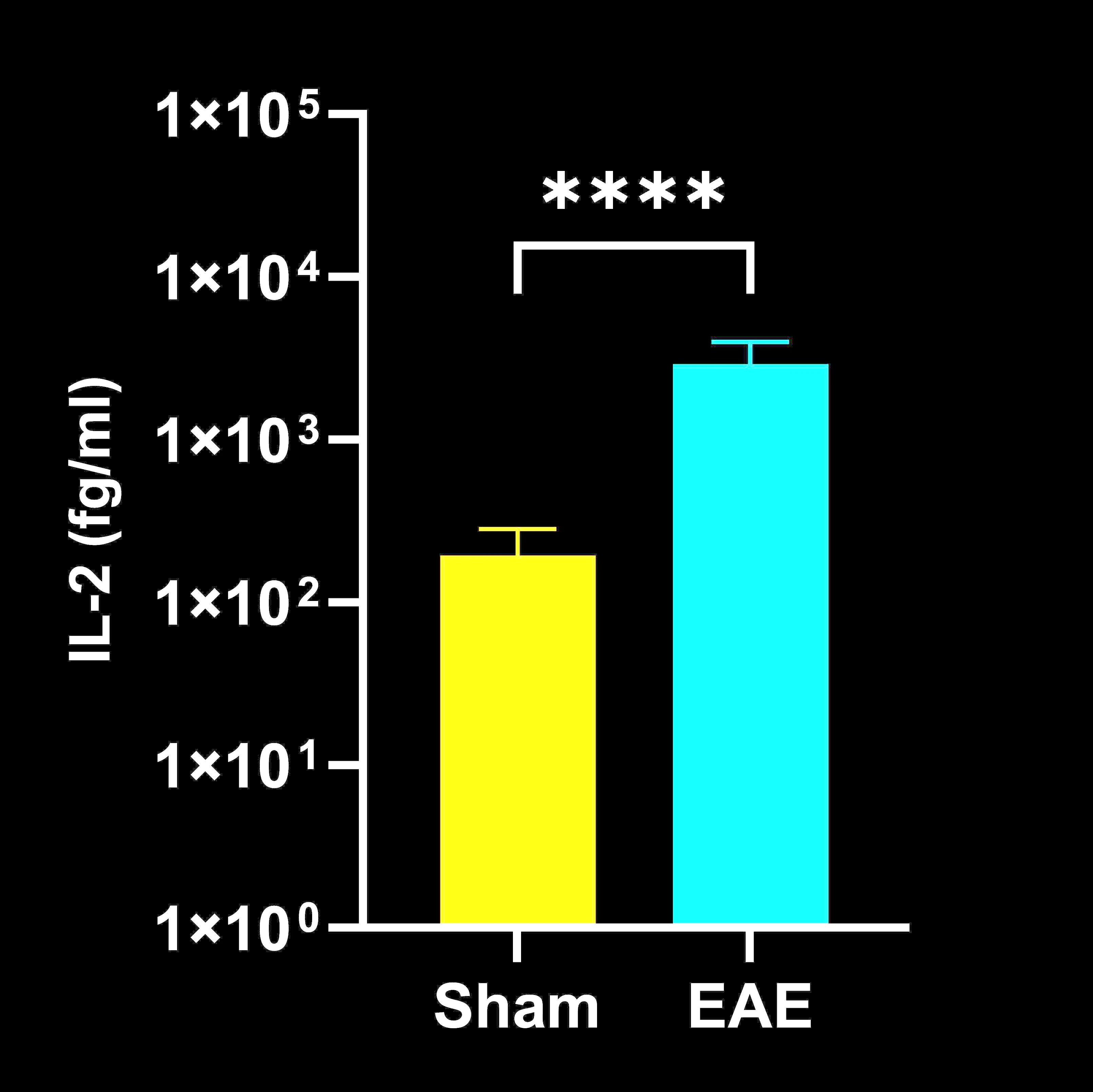
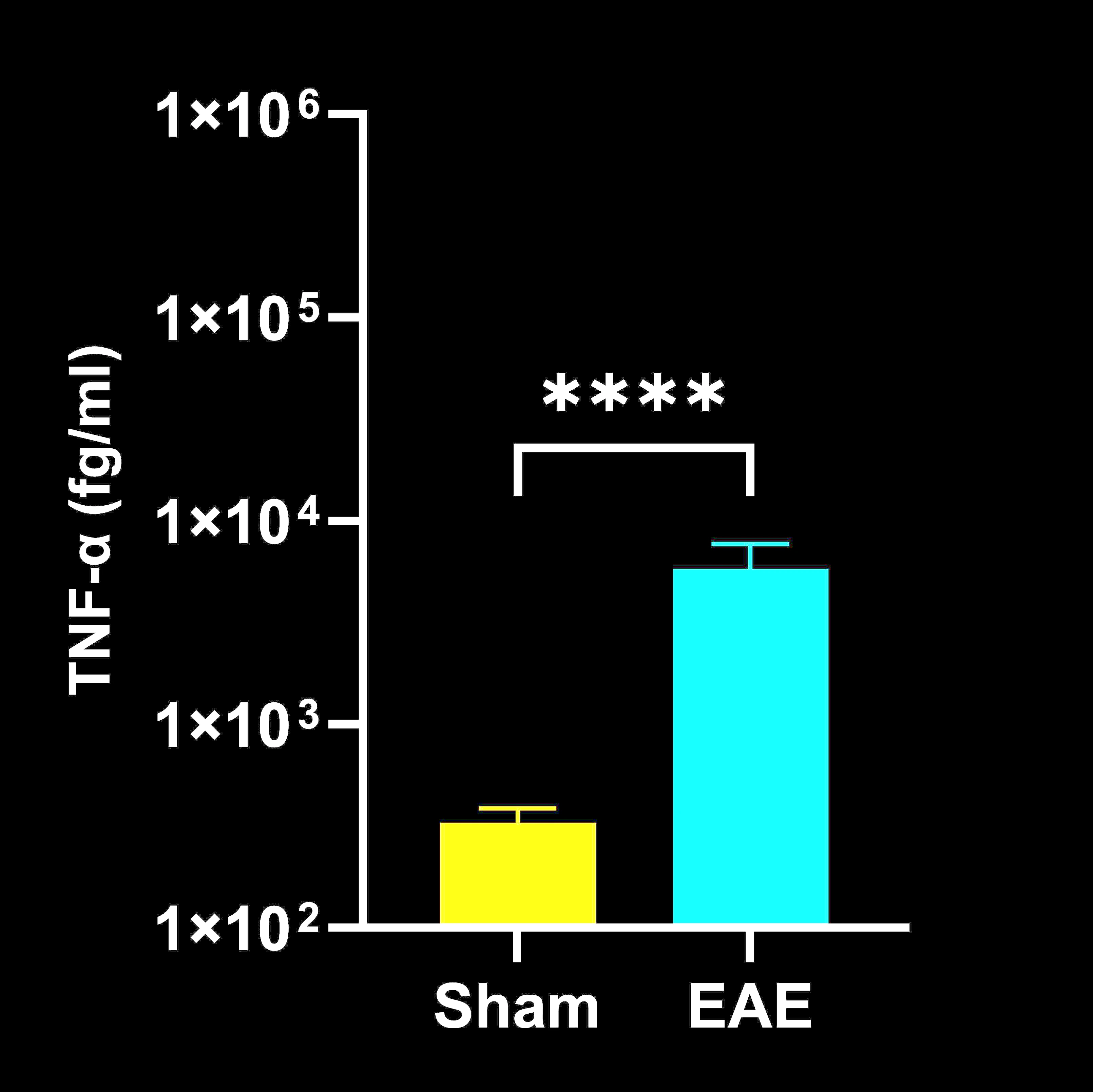
IL-2 and TNF-α were significantly elevated in the CSF, while IFN-γ, IL-1β, IL-2, IL-6, and TNF-α were increased in spinal cord homogenates; no significant differences were detected in the plasma. These findings indicate that inflammatory cytokine upregulation in EAE is largely confined to the CNS at the measured 21-day endpoint. Our observations are consistent with previous reports demonstrating increased cytokine levels in spinal cord tissue but not in serum (Alassiri, 2023), as well as selective changes in plasma cytokines, where some peak and subsequently decline over time (Borjini, 2016). This direct, multiple biological matrix cytokine analysis provides a complementary perspective to the more commonly reported clinical and pathological findings in EAE.
For an overview of our fluid and cell biomarker solutions, see: Fluid & Cell Biomarkers
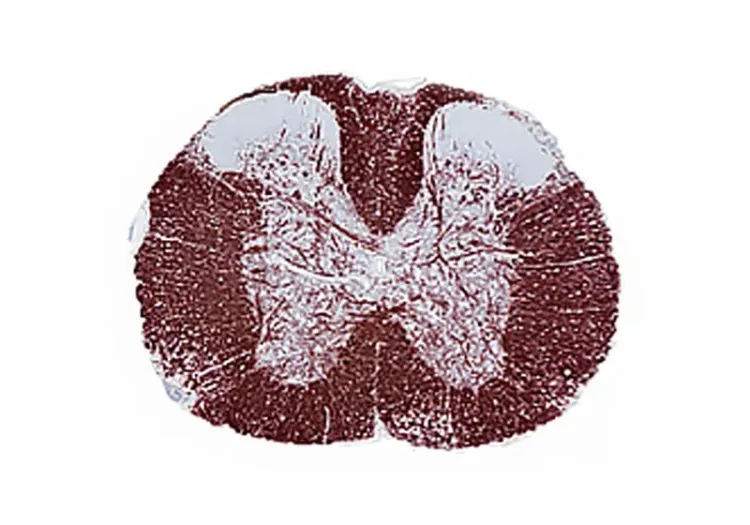
In the "Image Interactive" below, you can find results on microglia-neuron interactions, including high-resolution Multiplex Immunofluorescence tissue sections of spinal cords from the EAE mouse model and control mice.
How to use Our Interactive Viewer
Navigate through the “Image Story” via the left-hand panel or the on-screen arrows. You can pan around high-resolution microscopy images with your mouse, and zoom in/out using the scroll wheel or the +/- controls. The Control Panel (top-right) allows toggling of image channels and segmentation overlays. For the best experience, we recommend switching to full-screen mode. This Interactive Presentation enables you to explore the model’s neuropathology and associated functional deficits in detail, as if looking directly down the microscope.
Image Interactive describing cytokine analysis quantified by ultrahigh sensitivity ELISA in terminal plasma, CSF, and spinal cord homogenates, along with high-resolution multiplex immunofluorescence spinal cord tissue sections from the EAE mouse model.
Click to copy link
What Role do Cytokines Play in Experimental Autoimmune Encephalomyelitis (EAE)?
Multiple Sclerosis: Clinical and Pathological Features
Multiple sclerosis (MS) is a chronic neurodegenerative autoimmune disease of the central nervous system (CNS), characterized by demyelination and loss of oligodendrocytes. MS typically follows four clinical courses: relapsing-remitting MS (RRMS), primary progressive MS (PPMS), secondary progressive MS (SPMS), and progressive-relapsing MS (PRMS). Approximately 85% of patients initially present with RRMS, in which relapses of neurological dysfunction are followed by partial or complete remission. Symptoms vary widely and can include gait disturbances, visual impairment, and cognitive deficits.
T lymphocytes (CD3, red) in our EAE mouse model.
High magnification view of T lymphocytes (CD3, red) in the spinal cord in our EAE mouse model.
Role of T Cells in MS
A hallmark of MS pathology is the infiltration of autoreactive T lymphocytes, particularly CD4+ T helper (Th) cell subsets, into the CNS. Once recruited, Th cells secrete pro-inflammatory cytokines and chemokines that promote immune cell recruitment, activate resident glial cells, and contribute to blood–brain barrier (BBB) breakdown, demyelination, and axonal loss (Heng, 2022).
Among Th subsets, Th1 and Th17 cells are prominently involved. Th1 cells differentiate primarily under the influence of IL-12 and IFN-γ and drive macrophage activation and cell-mediated inflammation. Th17 cells arise in the presence of cytokines, such as IL-6 and TGF-β, and secrete IL-17A and GM-CSF.
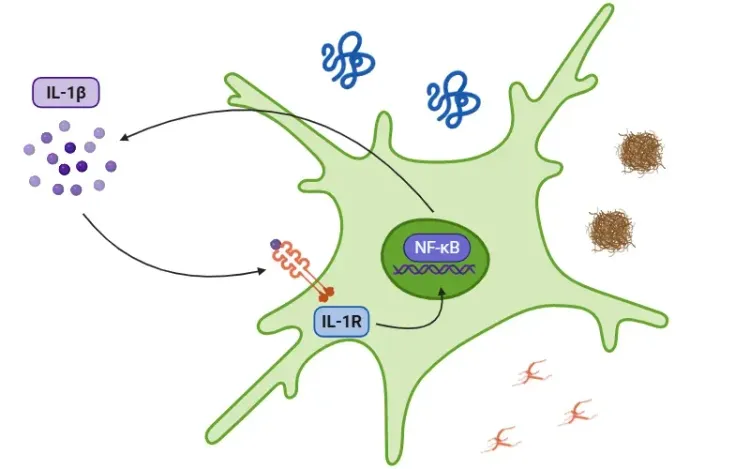
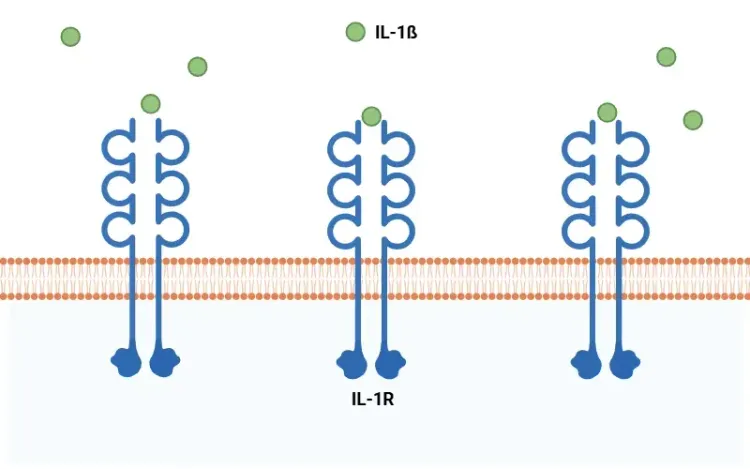
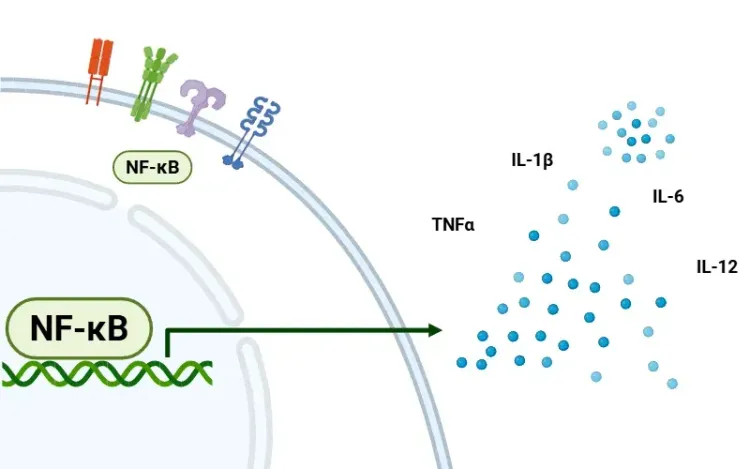
IL-17A is strongly linked to MS pathogenesis; signaling through the IL-17 receptor activates NF-κB, inducing additional pro-inflammatory cytokine production (including IL-1β) and sustaining recruitment of neutrophils and monocytes to inflammatory sites (Heng, 2022).
See our Resource: What is NF-κB (Nuclear Factor Kappa B)?
Glial Cells and NLRP3 Inflammasome
CNS-resident glial cells amplify the inflammatory response initiated by infiltrating T cells. Astrocytes are key contributors to MS immunopathology and respond to Th cell-derived cytokines by adopting a reactive phenotype that amplifies neuroinflammation and exacerbates tissue damage (Kunkl, 2022). Reactive astrocytes further compromise the BBB, and contribute to demyelination and axonal injury. Macrophages and microglia also become activated and release inflammatory mediators that sustain local immune responses and promote lesion development.
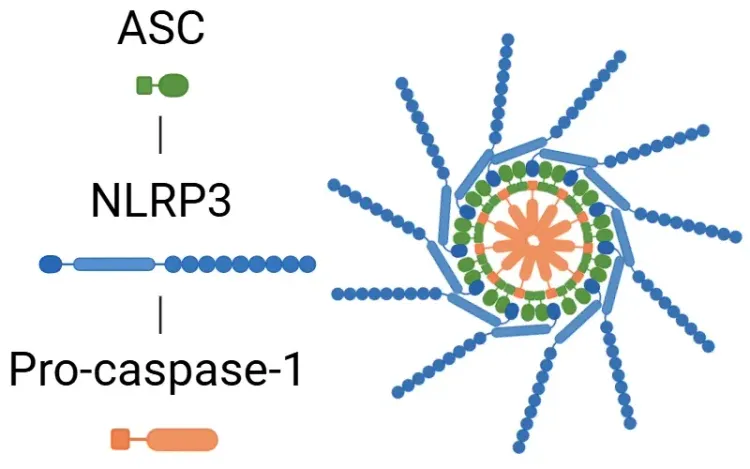
In addition, activation of the NLRP3 inflammasome represents an important innate immune mechanism in MS. The NLRP3 inflammasome is a multiprotein complex comprising the NLRP3 sensor protein, the ASC adaptor, and caspase-1. When activated, it triggers pyroptosis and the release of IL-1β and IL-18, thereby promoting neuroinflammation and neuronal damage (Xu, 2025). Consistent with this process, IL-1β levels are elevated in both MS and the EAE model (Borjini, 2016; Malhotra, 2020; Boraschi, 2023).
For more information, visit:
Astrocytes (GFAP, violet) demonstrating neuroinflammation in our EAE mouse model.
Macrophages/microglia (Iba-1, orange) demonstrating neuroinflammation in our EAE mouse model.
Cytokines and Chemokines in MS
Cytokines and chemokines are central mediators of immune-driven tissue damage in MS. Chemokines regulate leukocyte recruitment into the CNS, thereby promoting inflammation and demyelination (Arimitsu, 2025). Tumor necrosis factor-alpha (TNF-α) is a major pro-inflammatory cytokine in MS, elevated in the cerebrospinal fluid (CSF) of patients and in animal models (Borjini, 2016; Zahid, 2021). It is one of the most prominent cytokines in both MS and EAE lesions (Maguire, 2021). TNF-α contributes to secondary neuronal and axonal damage, though it may also exert context-dependent protective effects through distinct receptor pathways (e.g. via TNFR2 vs. TNFR1).
Beyond pro-inflammatory factors, immunoregulatory cytokines influence MS progression as well. For example, IL-2 and TGF-β drive naive T cell differentiation into regulatory T (Treg) cells, which maintain immune tolerance through inhibitory cytokine release and other mechanisms; an imbalance between pro-inflammatory Th17 cells and anti-inflammatory Treg cells is implicated in disease pathogenesis (Zhang, 2021). MS lesions are ultimately characterized by infiltrating T and B lymphocytes, activated macrophages/microglia, and a complex cytokine milieu that drives chronic inflammation, demyelination, and neuroaxonal degeneration.
To learn more about the role of cytokines in neurodegenerative diseases, see our Resources on: IL-1β & TNF-α
Experimental Autoimmune Encephalomyelitis as a Model of MS
Experimental Autoimmune Encephalomyelitis (EAE) is a gold-standard animal model of MS, recapitulating many pathologic features of the human disease. Cytokine-driven demyelination and neurodegeneration, central to MS pathology, are also key features of EAE. For example, IL-6 is increased in the brains of EAE mice (Leuti, 2021; Marin-Prida, 2022). Myelin oligodendrocyte glycoprotein (MOG)35–55-induced EAE in mice is widely used and reflects essential aspects of MS, including CNS immune cell infiltration, and, depending on the model, a relapsing-remitting or a chronic progressive disease course.
However, while many EAE studies focus on clinical, histopathological, and/or gene expression readouts, comparatively few have directly quantified cytokine concentrations across matched tissues and biofluids using highly sensitive multiplex immunoassays. This lack of data highlights the importance of multiple biological matrix cytokine profiling as a complement to traditional EAE endpoints.
See: What is EAE (Experimental Autoimmune Encephalomyelitis)? & Axonal Injury & Experimental Autoimmune Encephalomyelitis
EAE induction leads to hindlimb paralysis and neuroinflammation, with cytokine levels quantifiable across multiple biological matrices, including CSF, blood, and spinal cord.
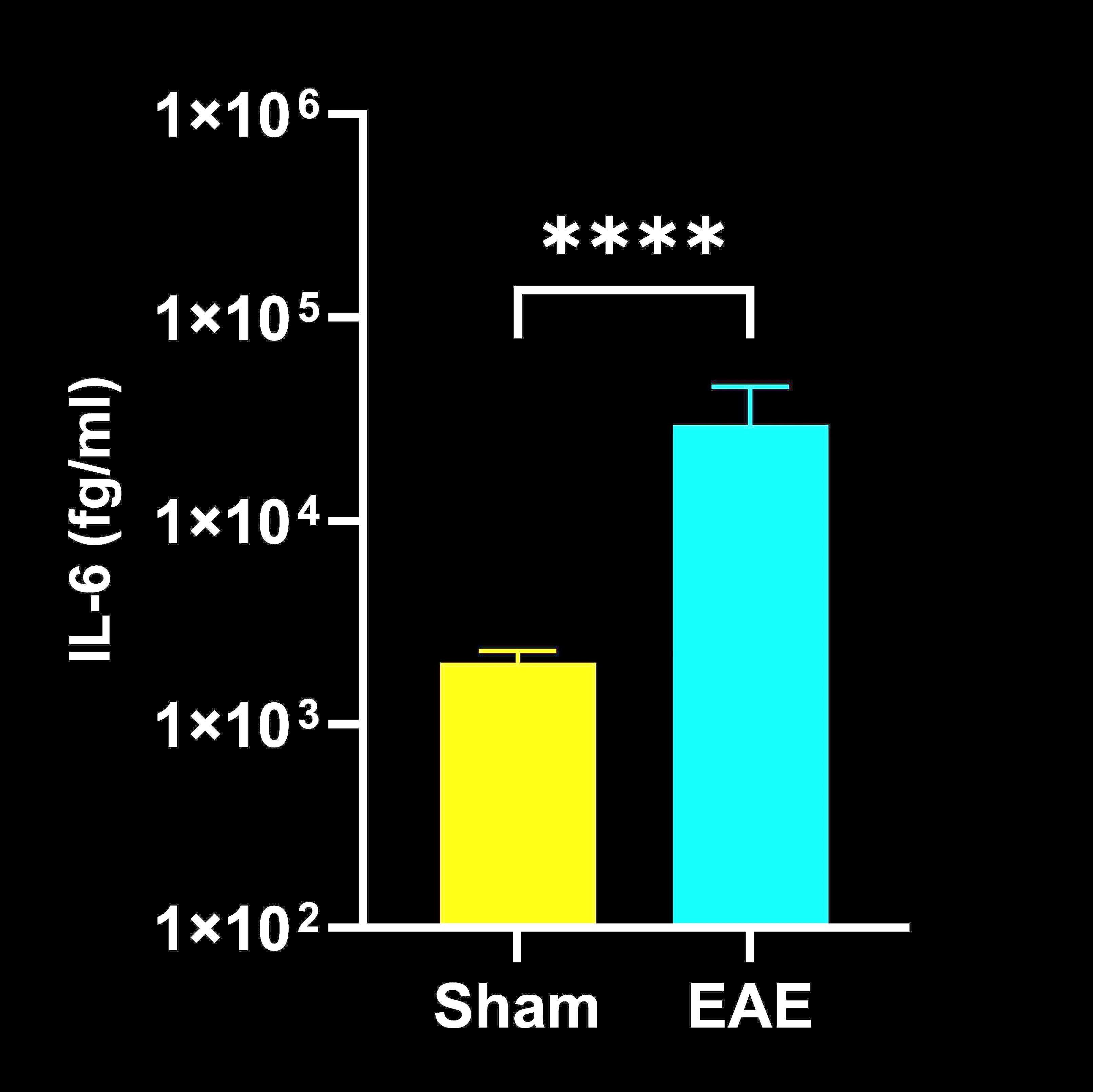
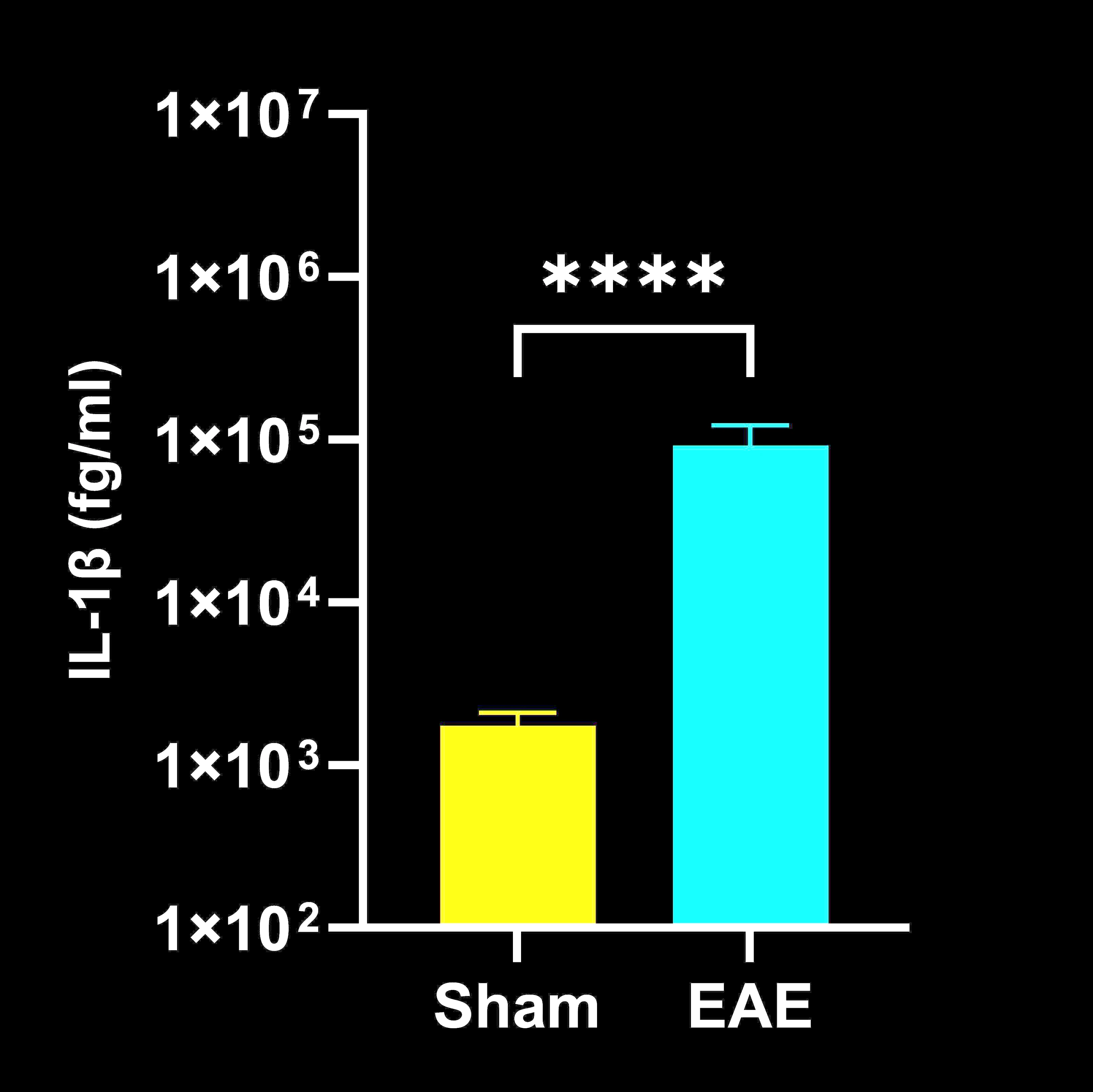
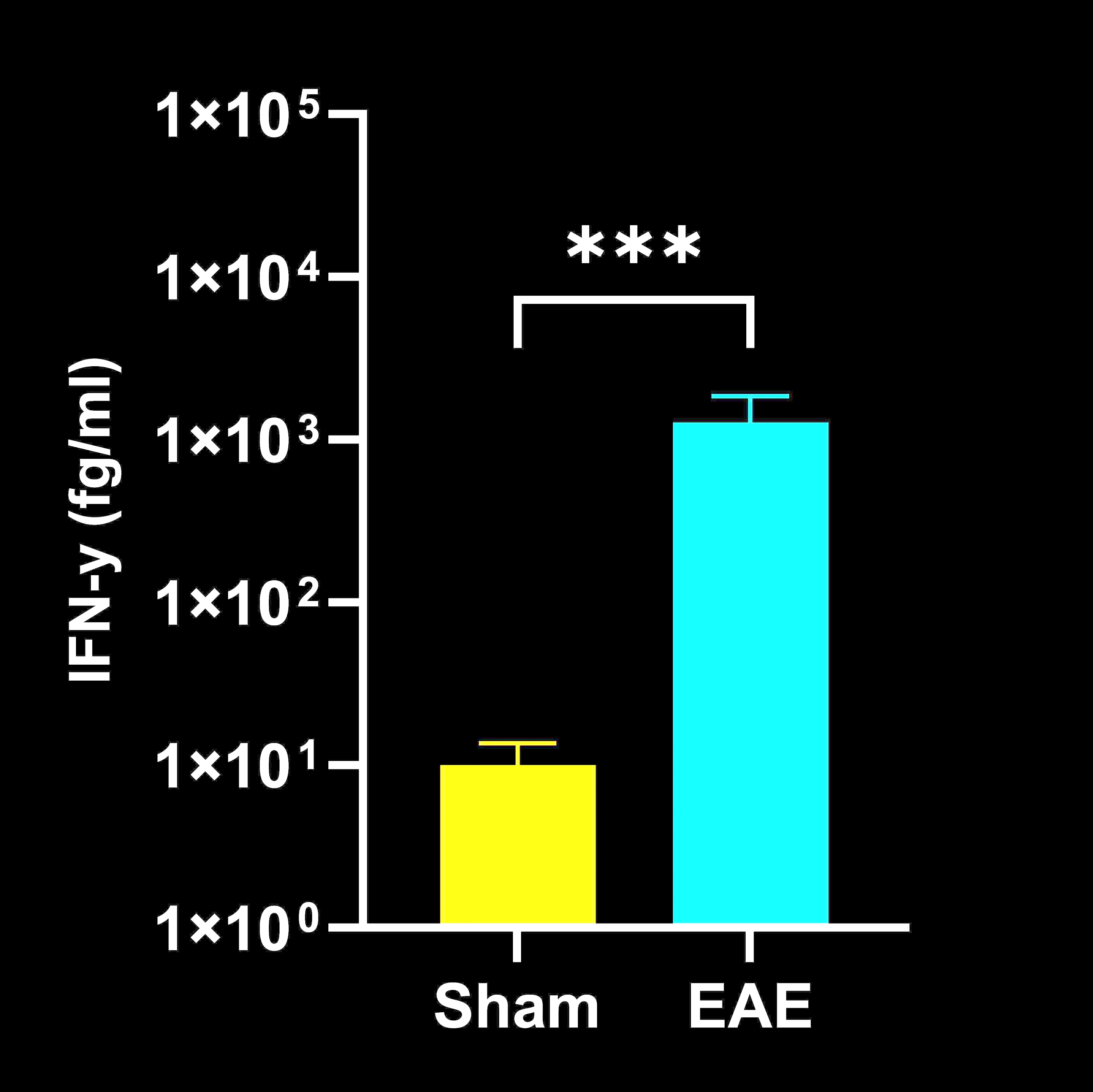
Cytokine Concentrations in Spinal Cord Homogenates
Spinal cord homogenate cytokine concentrations in EAE vs. Sham (control) mice; mean ± SEM, *** p<0.001; **** p<0.0001.
Cytokine Concentrations in Cerebrospinal Fluid (CSF)
Terminal CSF cytokine concentrations in EAE vs. Sham (control) mice; mean ± SEM, *** p<0.001.
Cytokine Concentrations in Plasma
Terminal plasma cytokine concentrations in EAE vs. Sham (control) mice; mean ± SEM.
FAQs
Discover more about our Neurodegenerative Diseases Models
Related Content
Up-to-date information on Cytokine Analysis and Neuroinflammation and best practices related to the evaluation of therapeutic agents in animal models of neurodegenerative diseases.
Experimental Autoimmune Encephalomyelitis (EAE) & Axonal Injury
This resource describes the methods available for measuring axonal damage & axon degeneration, including tissue markers and plasma & CSF neurofilament light chain (NfL; NF-L) levels, in the EAE model of multiple sclerosis (MS).
What is EAE (Experimental Autoimmune Encephalomyelitis)?
An overview of EAE animal models of multiple sclerosis (MS), including pathophysiology and utilization of positive controls for preclinical therapeutic studies.
Interleukin-1 Beta (IL-1β) and Neurodegenerative Diseases
The role of IL-1beta in neurodegenerative diseases, including Alzheimer's disease (AD), Parkinson’s disease (PD), and amyotrophic lateral sclerosis (ALS).
TNF-α (TNF-alpha) & Microglia in Neurodegenerative Diseases
An overview of the function of tumor necrosis factor-alpha (TNF-α) in microglia and its contribution to the progression of neurodegeneration.
NLRP3 Inflammasome and Neurodegenerative Diseases
An overview of the NLRP3 inflammasome and its role in neurodegenerative diseases, including Alzheimer's disease, Parkinson’s disease, and ALS.
What Is IL-1β (IL-1b)? Function, Signaling, and Biological Role
An overview of IL-1β, including its signaling pathways, involvement in disease mechanisms, and potential therapeutic targets.
What is NLRP3?
An overview of NLRP3 inflammasome activation triggers, disease associations, and therapeutic targeting strategies.
TNF-α & (TNF-alpha) Astrocytes in Neurodegenerative Diseases
An overview of TNF-α signaling in astrocytes, its role in neurodegeneration, and therapeutic strategies targeting this pathway..
What is NF-κB (Nuclear Factor Kappa B)?
An overview of NF-κB, highlighting its role in inflammation and diseases (including neurological disorders), and therapeutic strategies targeting NF-κB.
What is Pyroptosis? | A Drug Development Perspective
An overview of pyroptosis, its role in various diseases, and therapeutic strategies related to pyroptosis pathways.
Inflammasome – A Therapeutic Target for Multiple Diseases
An overview of inflammasomes, including their mechanisms of action, roles in diseases, and targeting for drug development.