Neuromuscular Junction (NMJ) Denervation in the TDP-43ΔNLS (rNLS8) Mouse Model of ALS
Biospective has industry-leading expertise in analysis of innervation, denervation, and other morphological changes in the neuromuscular junction (NMJ). Our services include high-quality tissue sectioning, multiplex immunofluorescence (mIF) staining, slide scanning, and quantitative image analysis of the NMJ in fixed muscle specimens from mouse, rat, and other animal models of neuromuscular and muscle diseases.
Explore our Services:
- What Services does Biospective offer for NMJ Staining & NMJ Analysis?
- What is Biospective's Process for NMJ Staining & Analysis?
- What is the Neuromuscular Junction (NMJ)?
- What is the Value of NMJ Analysis in Animal Models?
- How is the NMJ Affected in the TDP-43ΔNLS (rNLS8) Mouse Model of ALS?
- How does Automation improve Results?
What Services does Biospective offer for NMJ Staining & NMJ Analysis?
High-resolution NMJ detection, spatial mapping, innervation assessment, and morphological characterization for neuromuscular disease research.
The neuromuscular junction (NMJ) is affected in a wide variety of neurological and muscle diseases, including:
- Amyotrophic lateral sclerosis (ALS) (Verma, 2022)
- Myasthenia gravis (Vincent, 2002)
- Charcot-Marie-Tooth disease (Cipriani, 2018)
- Spinal muscular atrophy (SMA) (Murray, 2010) and
- Duchenne muscular dystrophy (DMD) (Pratt, 2015)
Quantitative analysis of NMJ features, such as NMJ innervation and NMJ denervation, is critical to understanding disease progression and response to therapeutic intervention, particularly with respect to ALS pathology and NMJ pathology in other neuromuscular disease models. Our team at Biospective has developed robust methods for multiplex immunofluorescence staining and quantitative image analysis of NMJs from muscle tissue sections, including optimization for ALS neuromuscular junction studies.
What is Biospective's Process for NMJ Staining & Analysis?
Well-established protocols for muscle sample preparation, staining, slide scanning, and quantitative image analysis.
Our Process for NMJ Staining & Analysis
At Biospective, we have implemented a standardized, highly reproducible multi-step process for staining and analysis of NMJs from formalin-fixed muscles:
1. Tissue Sectioning
- We embed the muscles in OCT before freezing and storage at -80oC
- We generate high-quality, fixed-frozen tissue sections using specially-equipped microtomes
- Sections are mounted on high-performance glass slides ensure adherence during the staining process
2. Multiplex Immunofluorescence NMJ Staining of Tissue Sections
- We perform multiplex staining for:
- Presynaptic terminal (SV2A)
- Motor endplates (alpha-bungarotoxin)
- Innervating axon (beta-III-tubulin)
- Staining is conducted using a high-throughput, automated IHC/IF instrument to ensure consistency and reproducibility
3. Slide Scanning
- Whole sections are digitized at ultrahigh spatial resolution using a digital slide scanner
- The resulting images are used for both visualization and quantitative analysis
4. Image Segmentation, NMJ Quantification, and NMJ Analysis
- Our imaging scientists have developed advanced, fully-automated segmentation methods for presynaptic terminals (SV2A), motor endplates (α-bungarotoxin), and innervating axons (β-III-tubulin)
- Our neuromuscular junction analysis tools allow for high-throughput, low bias neuromuscular junction quantification
- We derive a range of quantitative metrics from the segmented images to characterize NMJ morphology and functional state
Sample Collection, Preparation, and Shipping Guidelines
We provide comprehensive support to ensure sample integrity and data reliability:
- Sample Collection: Animals should be perfused with cold PBS and/or 10% neutral-buffered formalin, and the muscles should be carefully extracted.
- Sample Preparation: Muscle tissue must be briefly fixed in 10% neutral-buffered formalin and snap frozen.
- Sample Shipping: Samples must be shipped on dry ice using insulated containers, avoiding repeated freeze-thaw cycles.
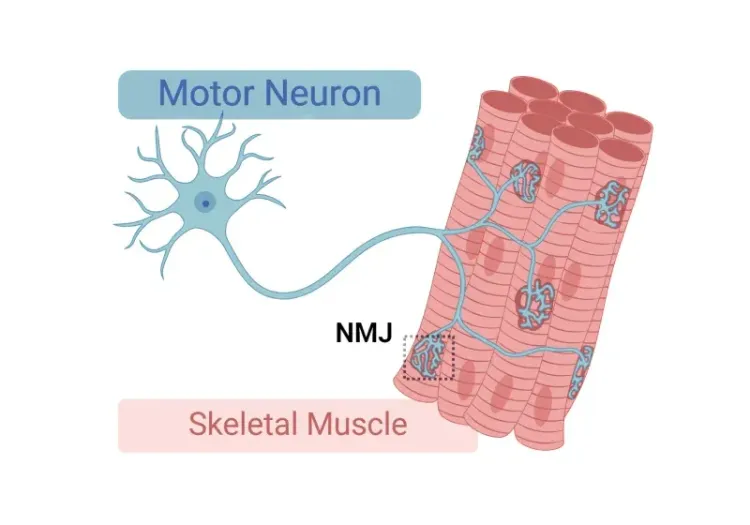
Schematic illustrating the key components of the neuromuscular junction (NMJ), including presynaptic motor neuron terminals, postsynaptic motor endplate, terminal Schwann cells, and kranocytes. Abbreviations: Neuromuscular Junction (NMJ), Acetylcholine (ACh), ACh Receptors (AChR).
What is the Neuromuscular Junction (NMJ)?
A brief overview of the NMJ and why robust quantitative analysis of NMJ pathology is important.
The neuromuscular junction (NMJ) is a specialized synapse that enables communication between motor neurons and skeletal muscle fibers, allowing precise muscle contractions. Presynaptic motor neuron terminals release acetylcholine (ACh) in response to electrical signals, which binds to receptors in the motor endplate of the muscle membrane, triggering an action potential and subsequent muscle contraction.
Why Analyze the NMJ?
- Early site of pathology: NMJs are affected before motor neuron loss in multiple neurodegenerative diseases (the “dying-back” process).
- Quantitative readout: Enables monitoring of denervation, remodeling, and therapeutic response in preclinical models.
- Experimentally accessible: The peripheral body location allows robust imaging & analysis, facilitating high-resolution assessment of synaptic integrity.
In this video, we provide an overview of our NMJ staining & analysis. It also includes an illustrative example of NMJ denervation in a mouse model of ALS, demonstrating how our services can be used to measure neurodegeneration, monitor disease progression, and evaluate potential therapies in preclinical studies.
Click to copy link
What is the Value of NMJ Analysis in Animal Models?
Evaluation of natural disease progression and response to therapeutic intervention in models of neurological and muscle diseases.
The NMJ is highly sensitive to innervation changes, morphological alterations, and degeneration across multiple neuromuscular diseases. NMJ analysis in animal models allows researchers to monitor disease progression and evaluate the efficacy of experimental therapeutic agents in preclinical studies.
Leveraging our validated imaging platforms, scientific expertise, and extensive experience, we provide robust, reproducible NMJ quantification across diverse models and species. Here, we highlight NMJ alterations in a mouse model of ALS that we routinely use for testing novel therapeutic interventions.
Amyotrophic Lateral Sclerosis (ALS) Models & ALS NMJ Pathology
An optimal animal model of ALS (or motor neuron disease [MND]) should exhibit the following key features:
- Symptom similarity to human ALS / MND, including motor deficits and NMJ vulnerability
- Progressive disease course, allowing longitudinal studies of pathology
- Disease modification, enabling interventions that alter disease progression
- Availability of age-appropriate animals for preclinical studies
- Low inter-animal variability, ensuring reproducible results across cohorts
TDP-43ΔNLS (rNLS8) Model
The TDP-43ΔNLS (rNLS8) Model mouse model meets these criteria, making it an attractive system for ALS drug development. This model expresses human TDP-43 with a defective nuclear localization signal (NLS), which impairs nuclear import and leads to cytoplasmic accumulation of TDP-43 in neurons. Over time, this mislocalized protein forms phosphorylated TDP-43 aggregates, recapitulating key pathological features observed in ALS patients.
At Biospective, we utilize both the original and modified versions of the rNLS8 ALS mouse model of TDP-43 proteinopathy:
- Original mouse model ("Off Dox"): rapid disease progression over weeks
- Biospective mouse model ("Low Dox"): slower, progressive disease over months
Both models exhibit progressive pathology, including:
- Motor neuron degeneration & regional brain atrophy
- Cytoplasmic TDP-43 accumulation and phosphorylated TDP-43 aggregates
- Motor deficits
- Brain, spinal cord, and neuromuscular junction (NMJ) pathology
For more information, see our Resources:
How is the NMJ Affected in the TDP-43ΔNLS (rNLS8) Mouse Model of ALS?
An illustrative example of NMJ denervation of altered morphology in a gold-standard TDP-43 mouse model of Amyotrophic Lateral Sclerosis (ALS).
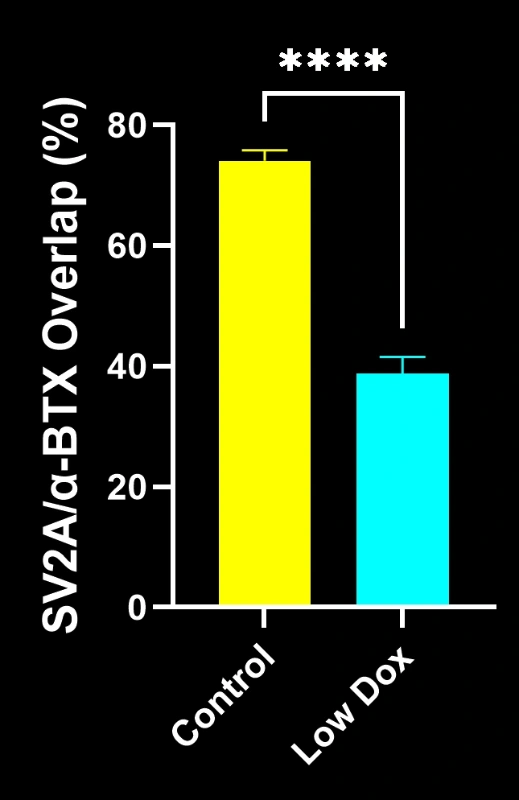
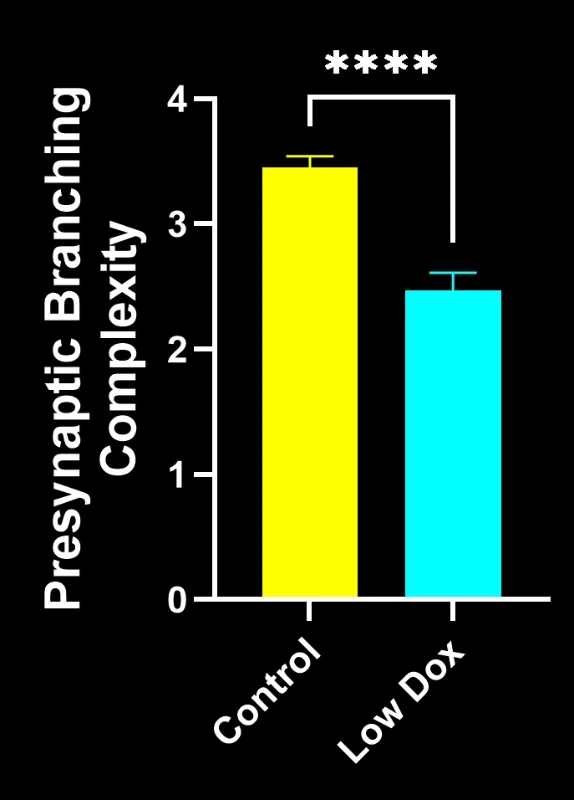
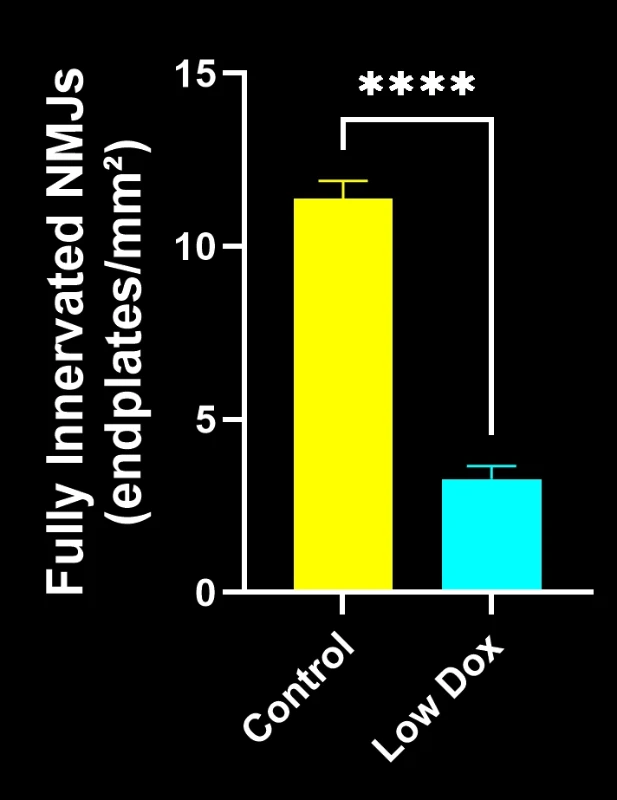
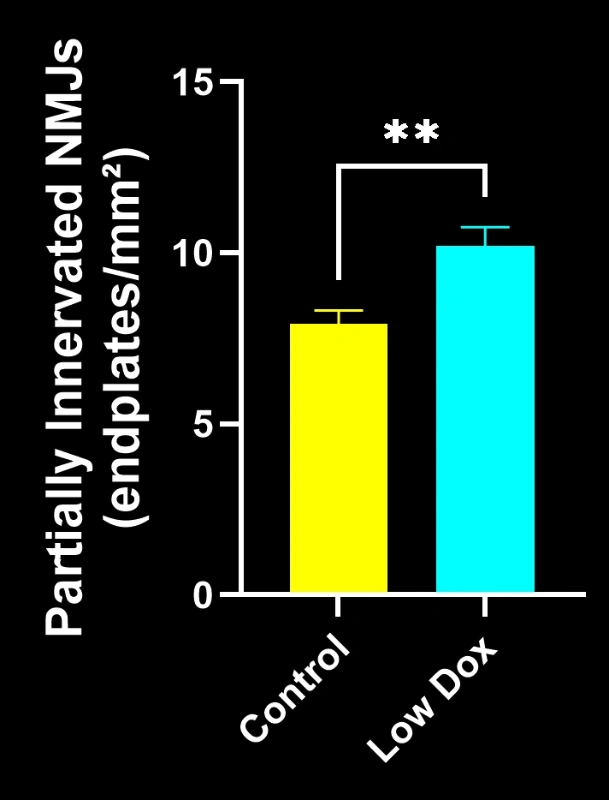
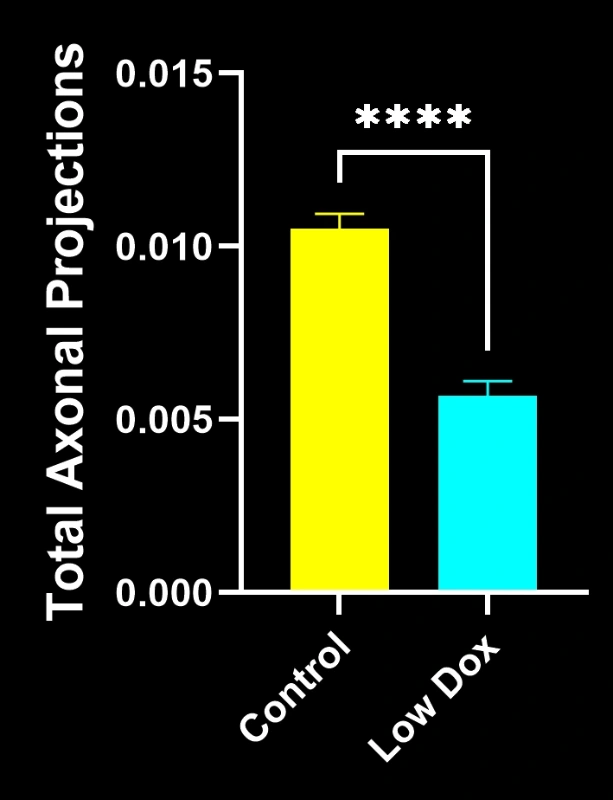
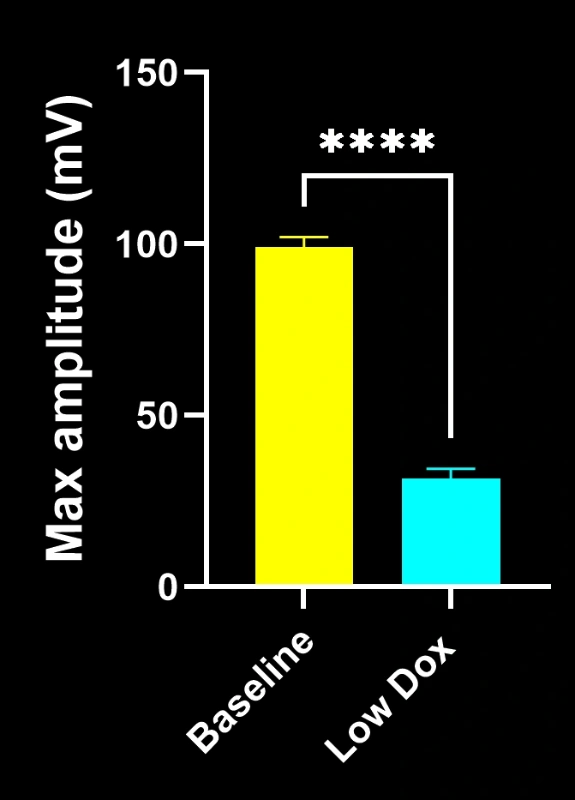
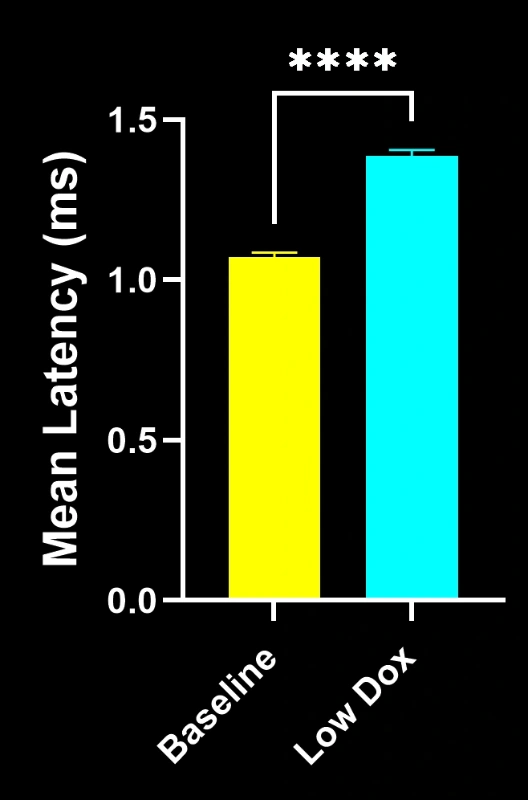
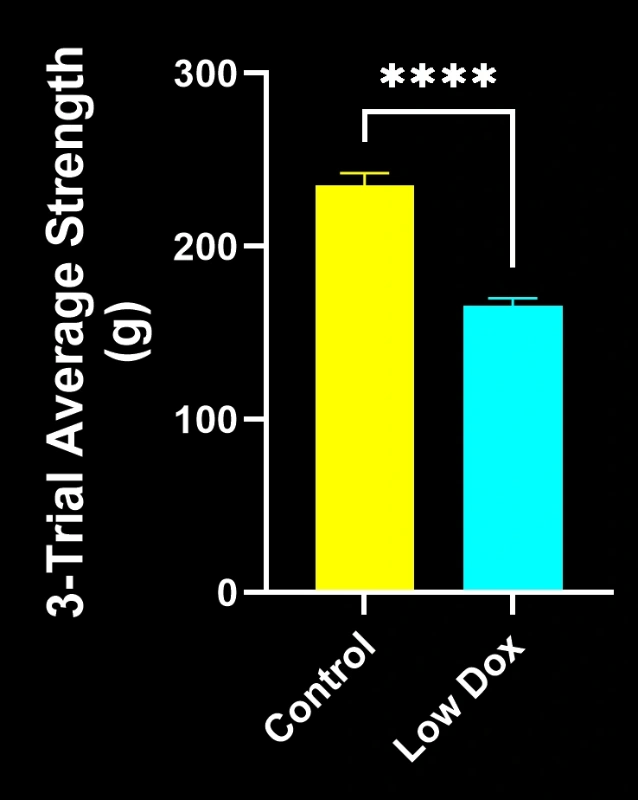
Biospective’s research scientists have conducted a rigorous evaluation of NMJ integrity and denervation in TDP-43 transgenic mouse models. This study compared tTA control mice with "Low Dox" rNLS8 mice at 8 weeks post-model induction.
Our study demonstrated:
- Marked reductions in SV2A/α-bungarotoxin co-localization, indicating NMJ denervation
- Decreased total axonal projections
- Simplified presynaptic architecture, reflecting ALS-like synaptic pathology
These findings highlight the progressive synaptic dysfunction at the NMJ and support this mouse model’s use for preclinical evaluation of therapeutic interventions targeting NMJ preservation in ALS.
In the "Image Interactive" below, you can find results from our NMJ analysis, including high-resolution Multiplex Immunofluorescence tissue sections of muscle from Biospective's “Low Dox” TDP-43ΔNLS (rNLS8) mouse model and control mice.
How to use Our Interactive Viewer
Navigate through the “Image Story” via the left-hand panel or the on-screen arrows. You can pan around high-resolution microscopy images with your mouse, and zoom in/out using the scroll wheel or the +/- controls. The Control Panel (top-right) allows toggling of image channels and segmentation overlays. For the best experience, we recommend switching to full-screen mode. This Interactive Presentation enables you to explore the model’s neuropathology and associated functional deficits in detail, as if looking directly down the microscope.
Image Interactive displaying NMJ analysis results, featuring high-resolution Multiplex Immunofluorescence muscle sections from Biospective’s “Low Dox” TDP-43ΔNLS (rNLS8) mouse model alongside control tissues.
Click to copy link
How does Automation improve Results?
The advantages of our automated staining & analysis approach over conventional manual methods.
We use automated neuromuscular junction staining and analysis with advanced high-throughput imaging and quantification platforms. This approach ensures accurate and and reproducible assessment of neuromuscular junction structure and integrity across multiple samples and experimental conditions.
Comparison between Manual and Automated Staining & Analysis of the NMJ
|
Feature |
Manual |
Automated |
|
Staining Consistency |
Variable antibody incubation and uneven reagent distribution |
Precisely controlled timing, temperature, and reagent application |
|
Throughput & Efficiency |
Time-consuming and limited number of slides analyzed |
High-throughput staining, imaging, and data processing |
|
Reproducibility |
Operator-dependent variability in staining and analysis |
Standardized protocols and algorithm-driven NMJ quantification |
|
Data Accuracy & |
Subjective NMJ assessment; limited quantitative precision |
Automated segmentation-localization, and morphological analysis |
|
Morphological Profiling |
Manual measurement of NMJ size, shape, and complexity |
Automated extraction of NMJ area, perimeter, branching, and complexity metrics |
|
Spatial Mapping |
Limited to selected regions-of-interest (ROIs) |
Whole-section mapping of NMJ distribution and synaptic coverage |
This table compares manual and automated neuromuscular junction (NMJ) staining and analysis across key criteria, including staining consistency, throughput and efficiency, reproducibility, data accuracy and analysis depth, morphological profiling, and spatial mapping.
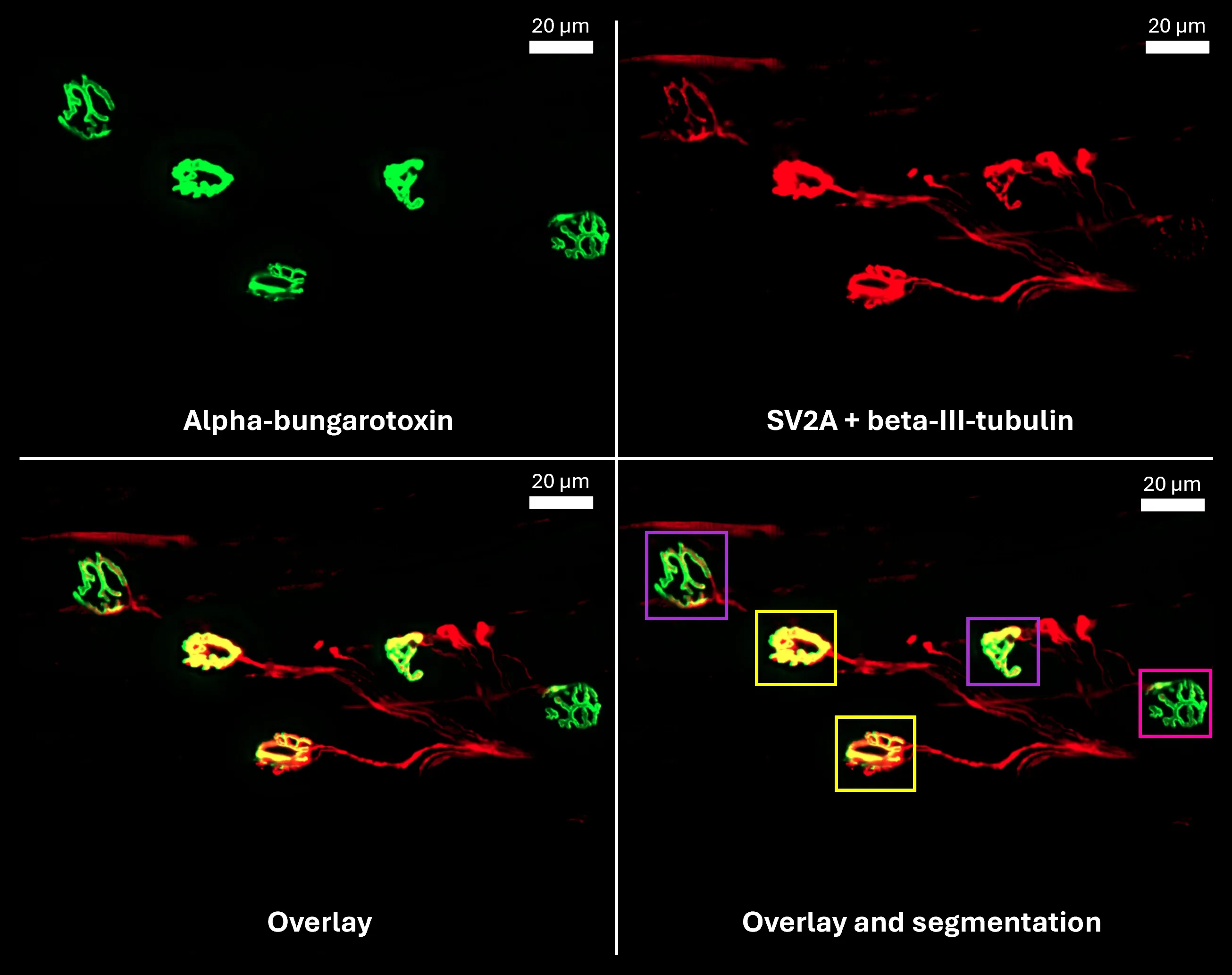
Example images of NMJs from mouse muscle that have undergone automated staining and image analysis using the processes developed at Biospective.
To discuss your study requirements or request a quote for Neuromuscular Junction (NMJ) staining and quantification services
Related Content
Up-to-date information on NMJ Staining and Analysis services.
Neuromuscular Junction (NMJ) Morphology & ALS Models
Insights into neuromuscular junction (NMJ), its role in amyotrophic lateral sclerosis (ALS), and tools & methods used to study morphological changes in NMJs.
A Guide to ALS Models for Drug Discovery
A Resource for the most effective use of research animal models (mouse & rat models) of Amyotrophic Lateral Sclerosis (ALS) for preclinical testing of therapeutics.
ALS Mouse Models & Spinal Motor Neurons
An overview of the involvement of spinal motor neurons in disease progression in mouse models of Amyotrophic Lateral Sclerosis (ALS).
TDP-43 ΔNLS (rNLS8) Mice for ALS Drug Development
This resource provides information about the use of the ΔNLS (deltaNLS, hTDP-43ΔNLS, hTDP-43DeltaNLS, dNLS, TDP43 NLS, rNLS8) TDP-43 transgenic mouse model of ALS for preclinical therapeutic studies.