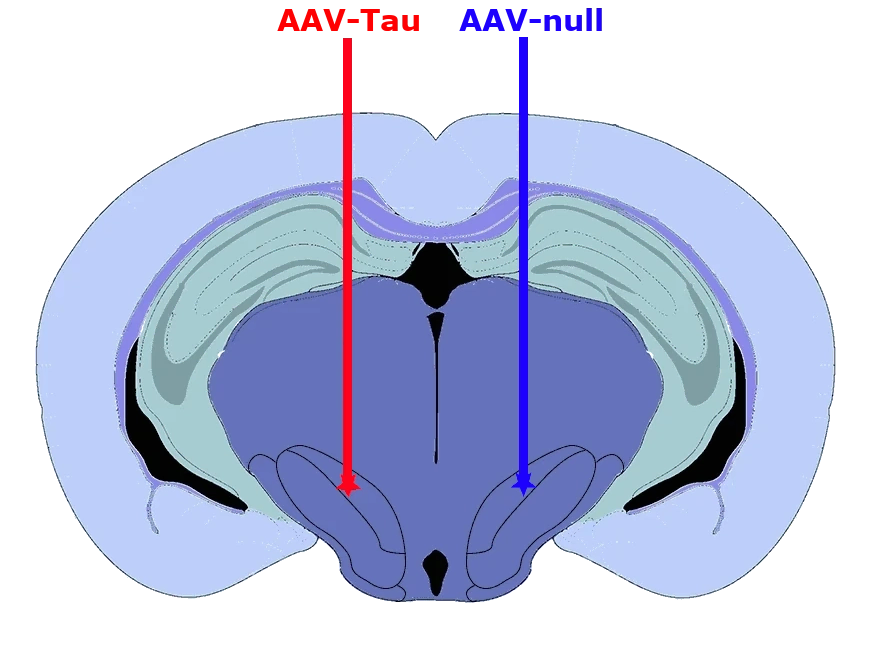
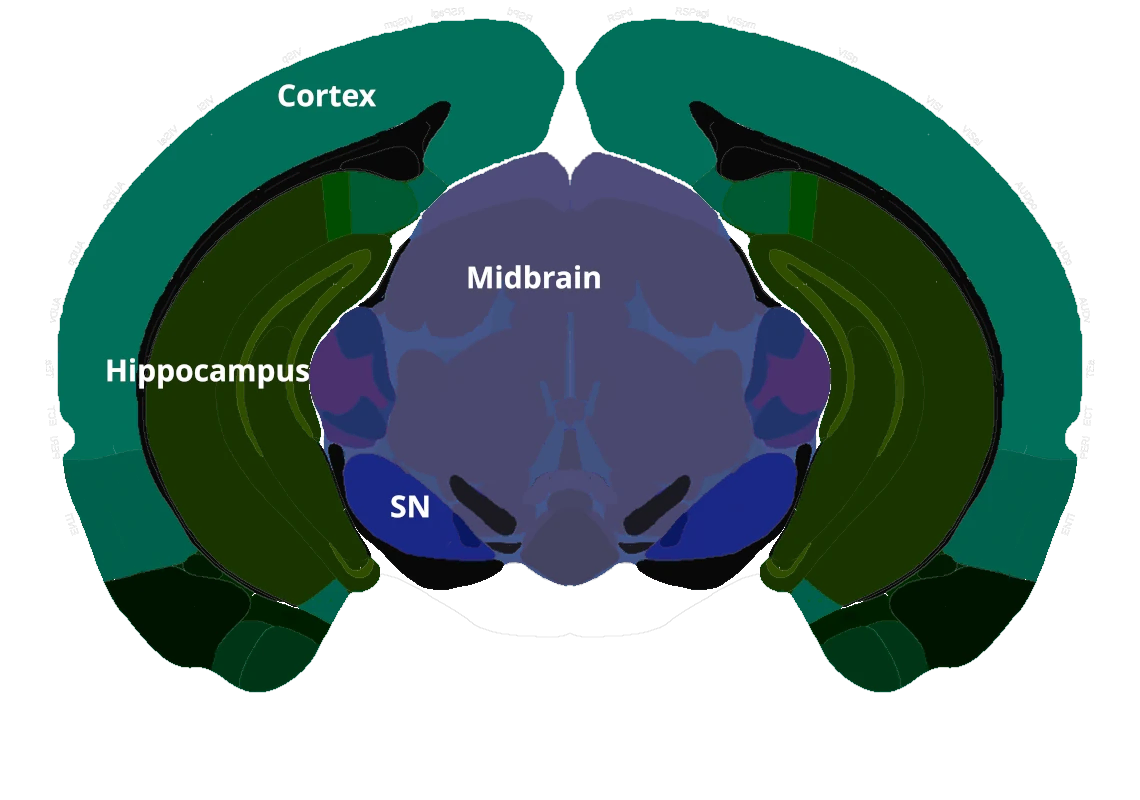
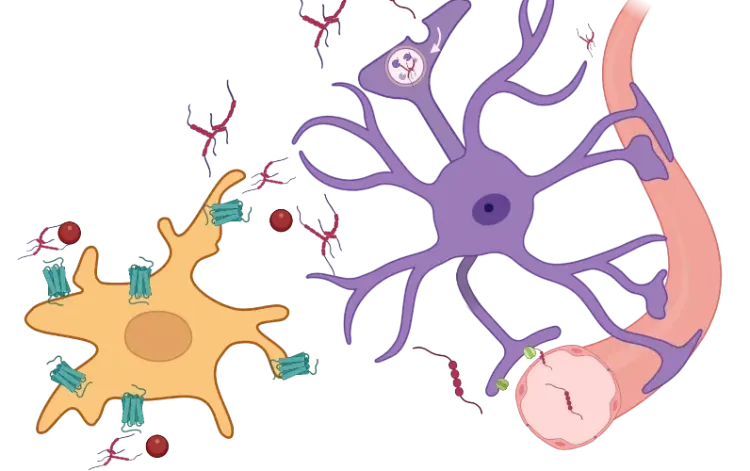
Characterization of a Novel AAV-hTau Mouse Model of Tauopathies with Parkinsonian Features
Biospective has industry-leading expertise in providing tau histology services, including multiplex immunofluorescence (mIF) staining using a variety of antibodies. We have unique capabilities for measurements of tau pathology features (e.g. aggregation, phosphorylation, conformation, cleavage) and spatial analysis of the complex relationships with other pathology (e.g. Aβ plaques) and associated neuroinflammation, including activated microglia and reactive astrocytes.
Table of Contents
What Contract Research Services does Biospective Offer for Tau Staining & Analysis?
Multiplex immunofluorescence staining, segmentation, morphological, and spatial analysis of tau in tissue sections from rodent models of Alzheimer’s disease and other tauopathies.
Biospective provides end-to-end characterization of tau aggregates and the glial responses using advanced multiplex immunofluorescence (mIF) tissue staining, high-resolution whole-slide imaging, and automated machine learning and deep learning based morphology analysis.
Our Tau Staining & Image Analysis Capabilities
Tau Staining & Multiplexing
We provide IHC/mIF staining using a wide variety of antibodies, including:
Non-Phosphorylated Tau: Tau15-25, Tau1-100, HT7
Phosphorylated Tau: AT8, PHF1, CP13, pTau-217
Cleaved Tau: N368, Asp421
Disease Conformation: MC-1
Amyloid Fibril Structure: pFTAA
We are continually developing protocols for other antibodies and we have excellent capabilities for implementation of custom markers.
To generate a more complete picture of the pathologic processes in tissue sections, we have extensive expertise in staining with multiplex IF panels.
Image Quantification & Analysis
We perform whole slide IHC/mIF stain density quantification and regional burden within specified regions-of-interest.
We can also perform complex spatial analysis to probe the relationships between misfolded proteins, neuroinflammation, and neurodegeneration.
What is Biospective’s Workflow for Staining & Quantitative Analysis of Tau?
Well-established protocols for brain sample preparation, staining, slide scanning, and quantitative image analysis.
Our Process for Tau Staining & Analysis
At Biospective, we have implemented a standardized, highly reproducible multi-step process for staining and analysis of tau from formalin-fixed brains:
Sample Preparation
High-precision microtome sectioning or cryosectioning of FFPE or fixed-frozen brains.
Custom antigen retrieval protocols optimized for each tau-specific antibody, ensuring high-affinity binding and preservation of tau morphology. Retrieval conditions are further customized for any additional antibodies included in the multiplex panel. We routinely perform heat-induced retrieval (HIER), enzymatic retrieval, formic acid retrieval, or a combination of these methods.
Stringent quality control (QC) of staining quality and specificity as well as tissue integrity.
Staining (IHC or Multiplex IF)
Tau Markers
Pathology Microenvironment Markers
Microglia (Iba-1 & other microglial markers)
Astrocytes (GFAP)
Neurons (NeuN; neuronal subtype markers, e.g. TH for dopaminergic neurons)
Other misfolded proteins (e.g. amyloid-β plaques, α-synuclein, TDP-43)
Subcellular & biochemical markers (e.g. lysosomes, autophagy, mitochondria, neurodegeneration)
DAPI (nuclei)
Advantages of Multiplexing
Multiplexing enables cell-type–specific analysis of the microenvironment on a single slide, accurately characterizing the cellular landscape surrounding individual plaques.
Imaging
Whole-section multichannel fluorescence scanning
Quantitative Analysis
We have developed fully-automated quantitative analysis for multiplex immunofluorescence to complement tau analysis, including amyloid-beta plaque segmentation and counting, glial cell morphology analysis, and microenvironment analysis.
Illustration of Biospective's process of collecting brain tissue samples from animal models, performing tissue sectioning, multiplex immunofluorescence staining, whole slide scanning, and quantitative image analysis.
Sample Collection, Preparation, and Shipping Guidelines
We provide comprehensive support to ensure sample integrity and data reliability:
Sample Collection: Animals should be perfused with cold PBS and/or 10% neutral-buffered formalin, and the brains should be carefully extracted.
Sample Preparation: Brains must be briefly properly fixed in 10% neutral-buffered formalin.
Sample Shipping: Samples must be shipped in PBS with sodium azide.
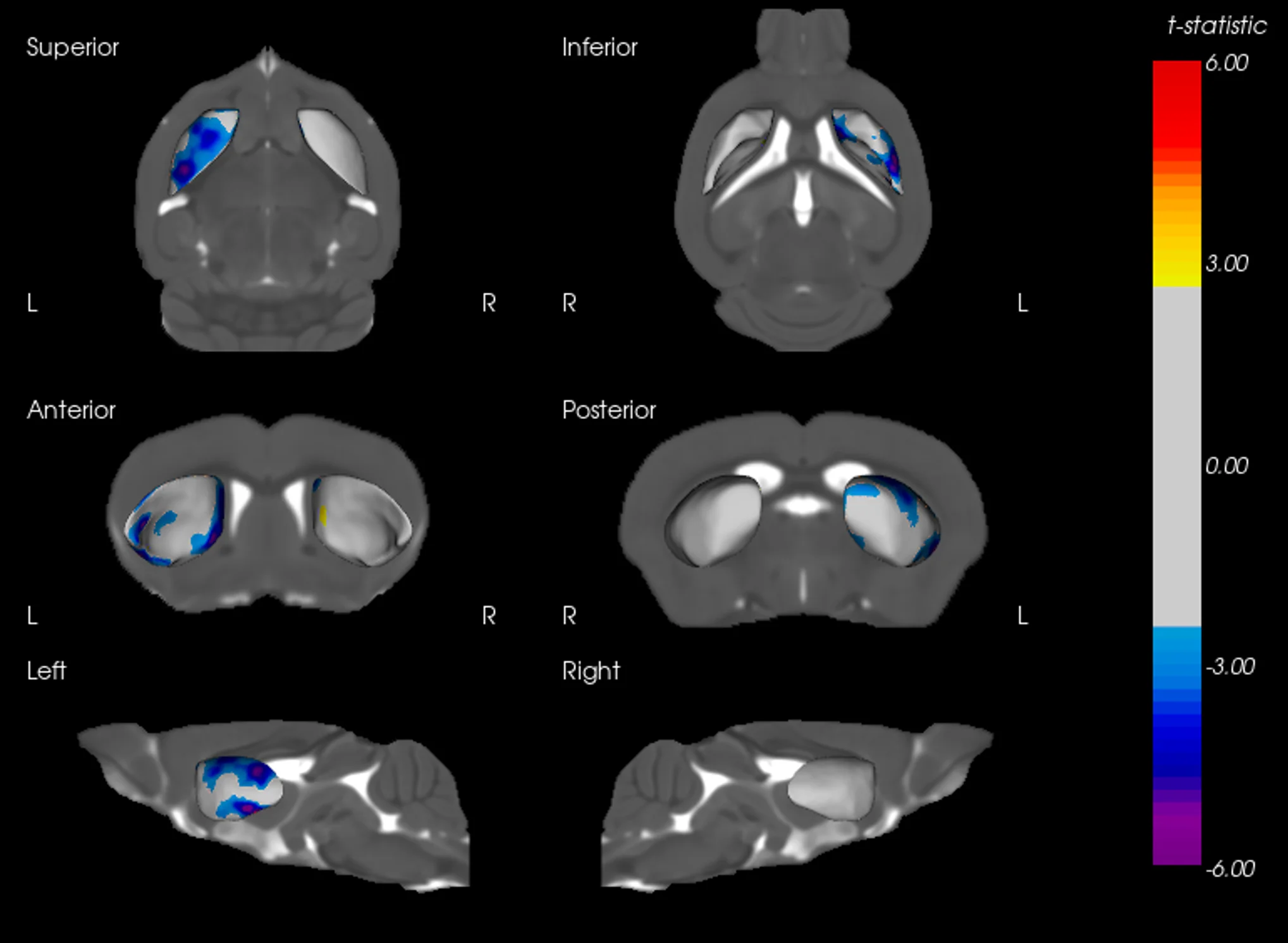
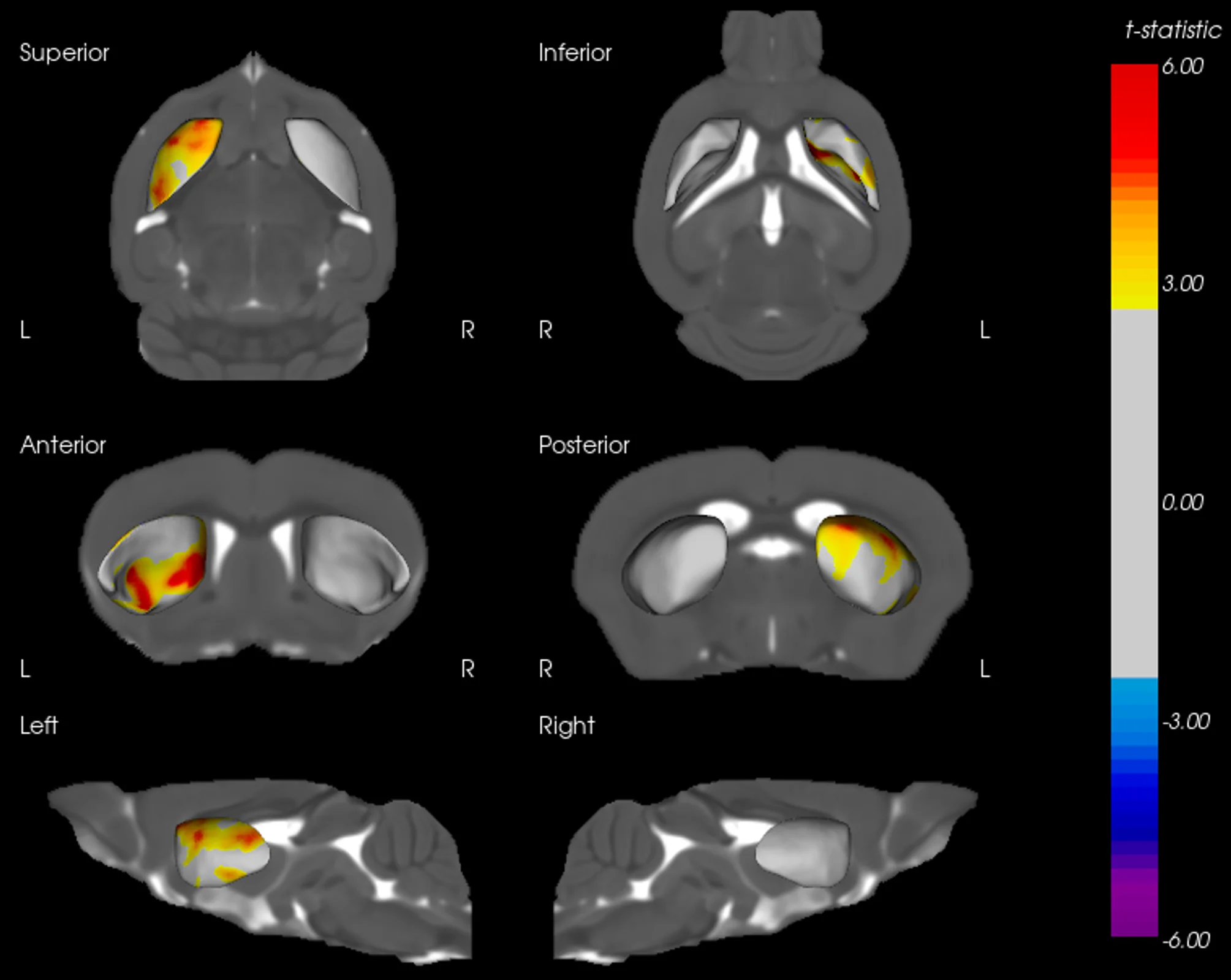
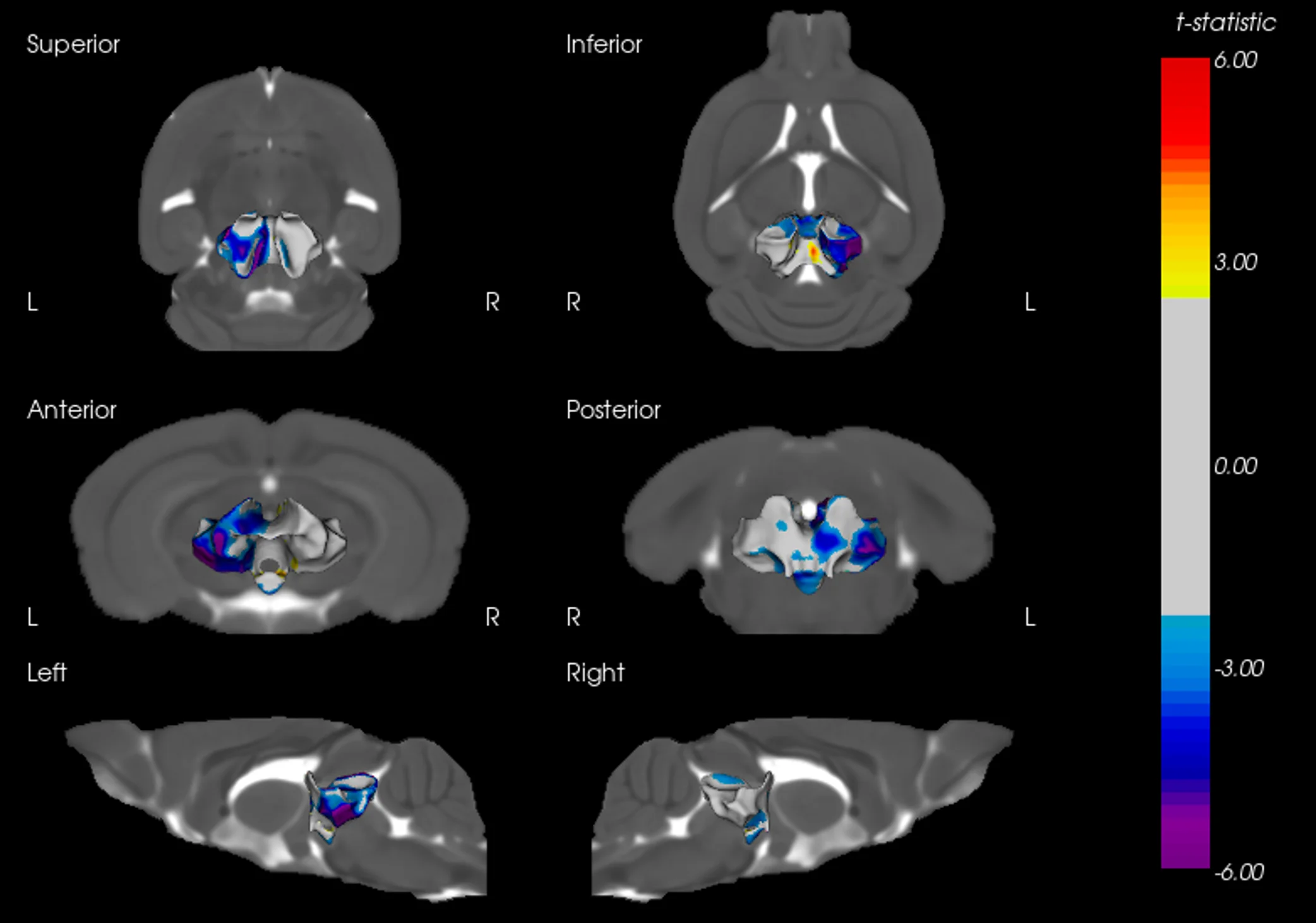
Example of Tau mIF Staining & Analysis in a Mouse Model of 4R Tauopathy
An illustrative example of tau pathology, neuroinflammation, and neurodegeneration in a mouse model of PSP & CBD.
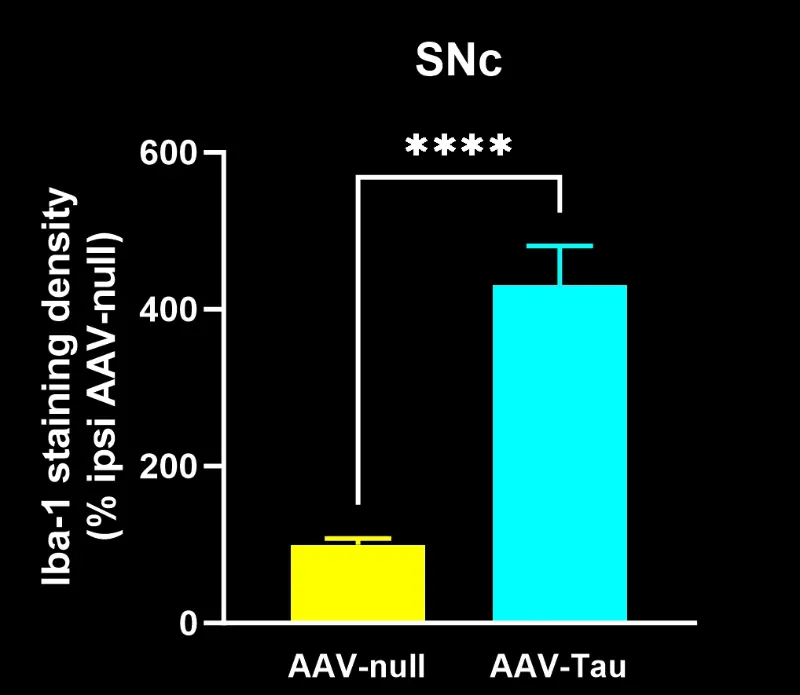
Biospective has developed a unique mouse model of 4R Tauopathy, which includes dopaminergic dysfunction and motor deficits. We have extensively validated this model using a broad range of in vivo and ex vivo assays, including various translational biomarkers to serve as a platform for the evaluation of therapeutic agents. We have leveraged our portfolio of tau markers and multiplex IF panels to characterize the neuropathology in these mice.
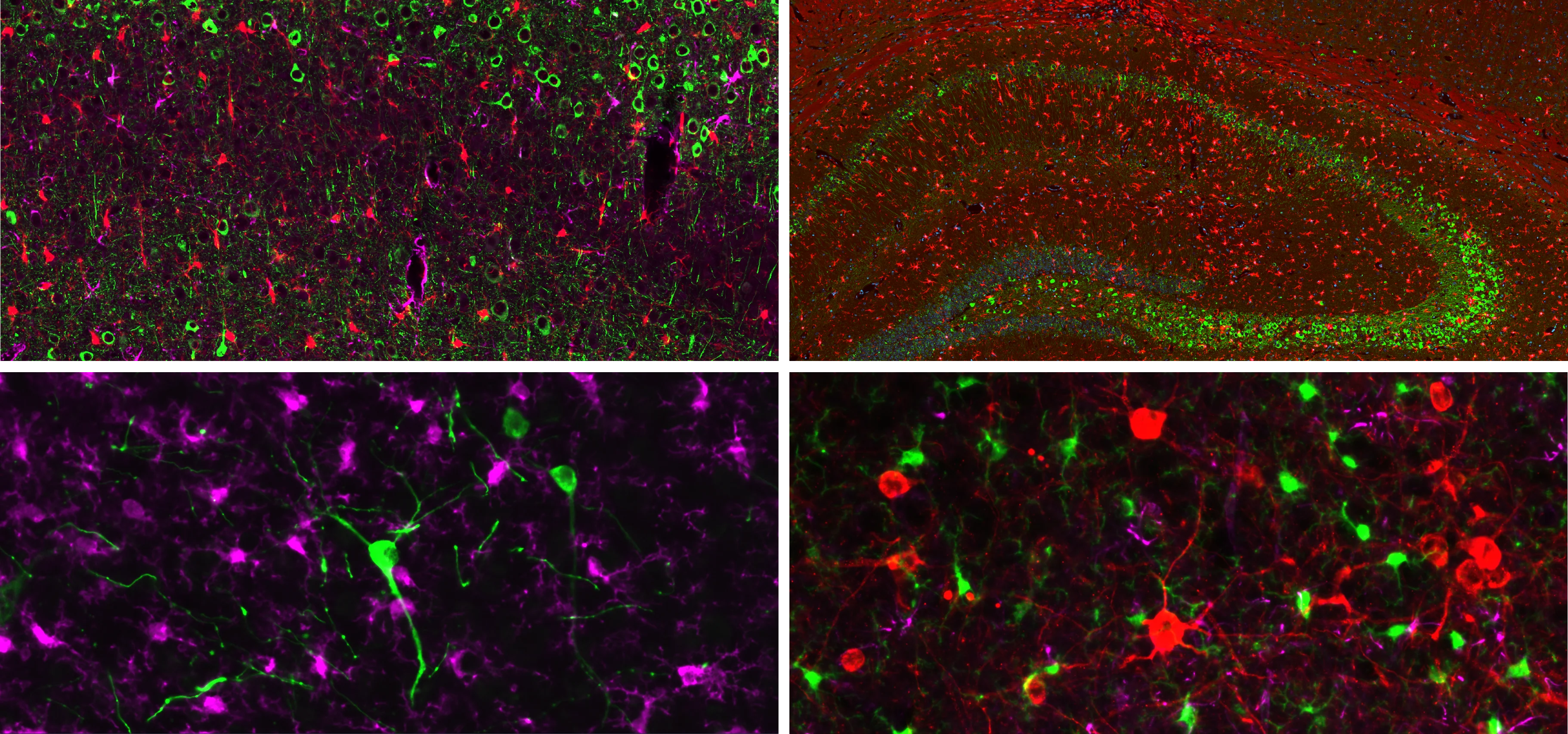
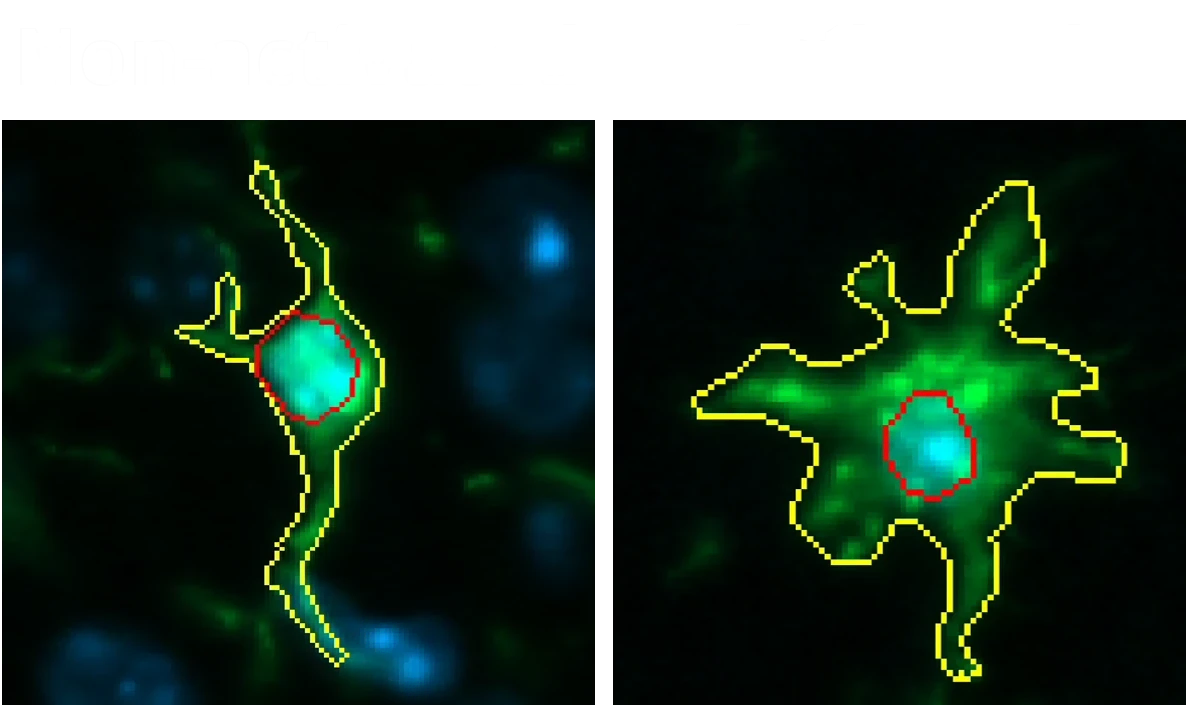
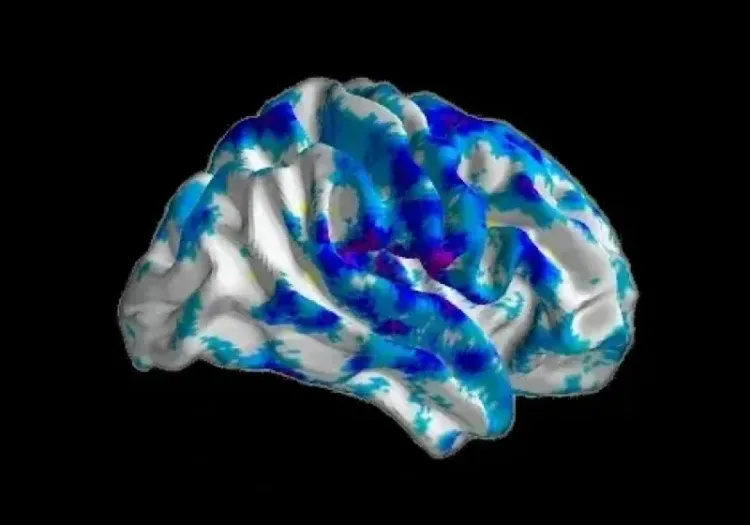
Representative multiplex immunofluorescence images of intracellular neuronal tau stained with various antibodies for phosphorylated and/or cleaved tau in mouse brain tissue sections. Microglia and astrocytes are also stained in these images.
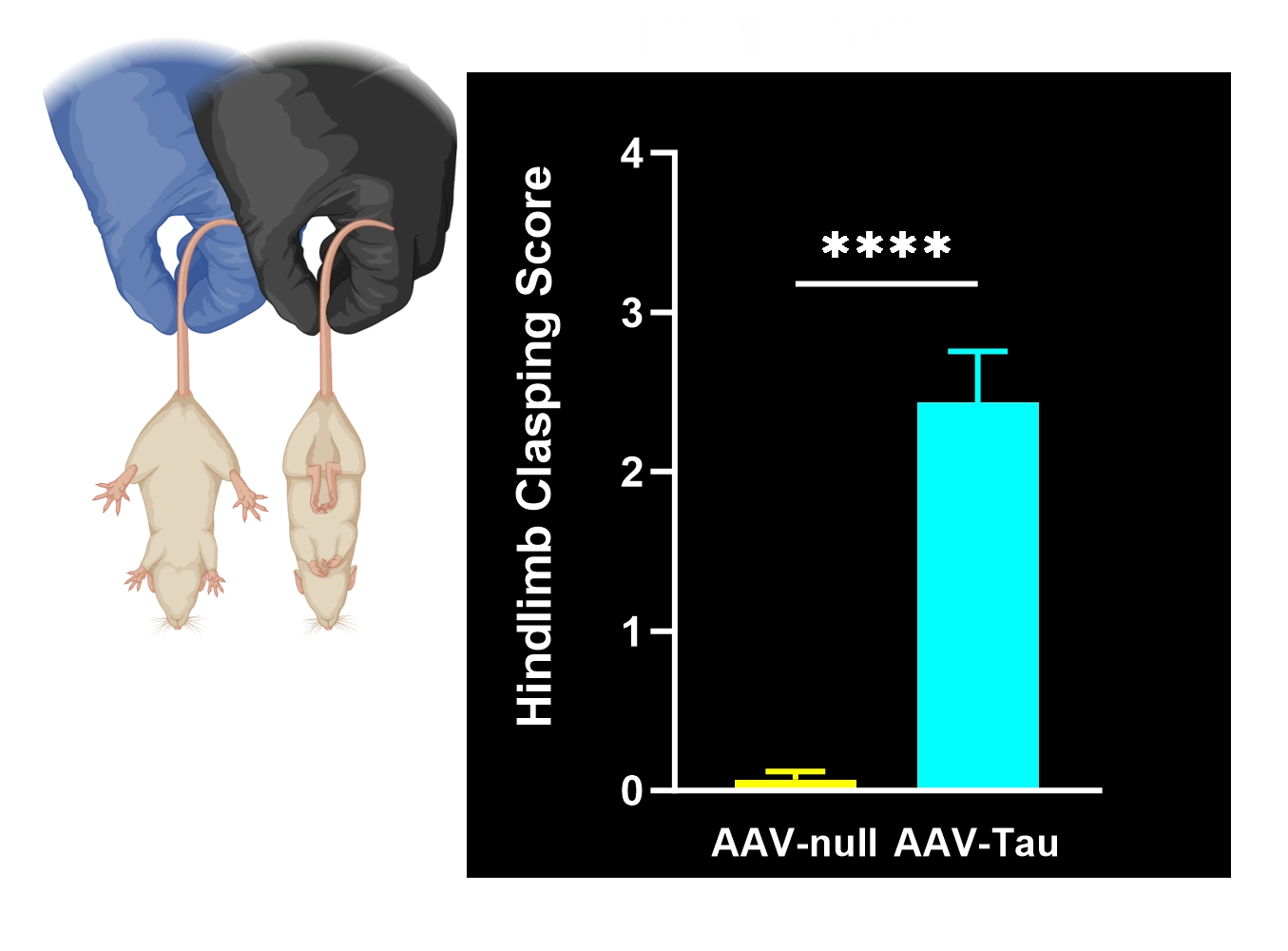
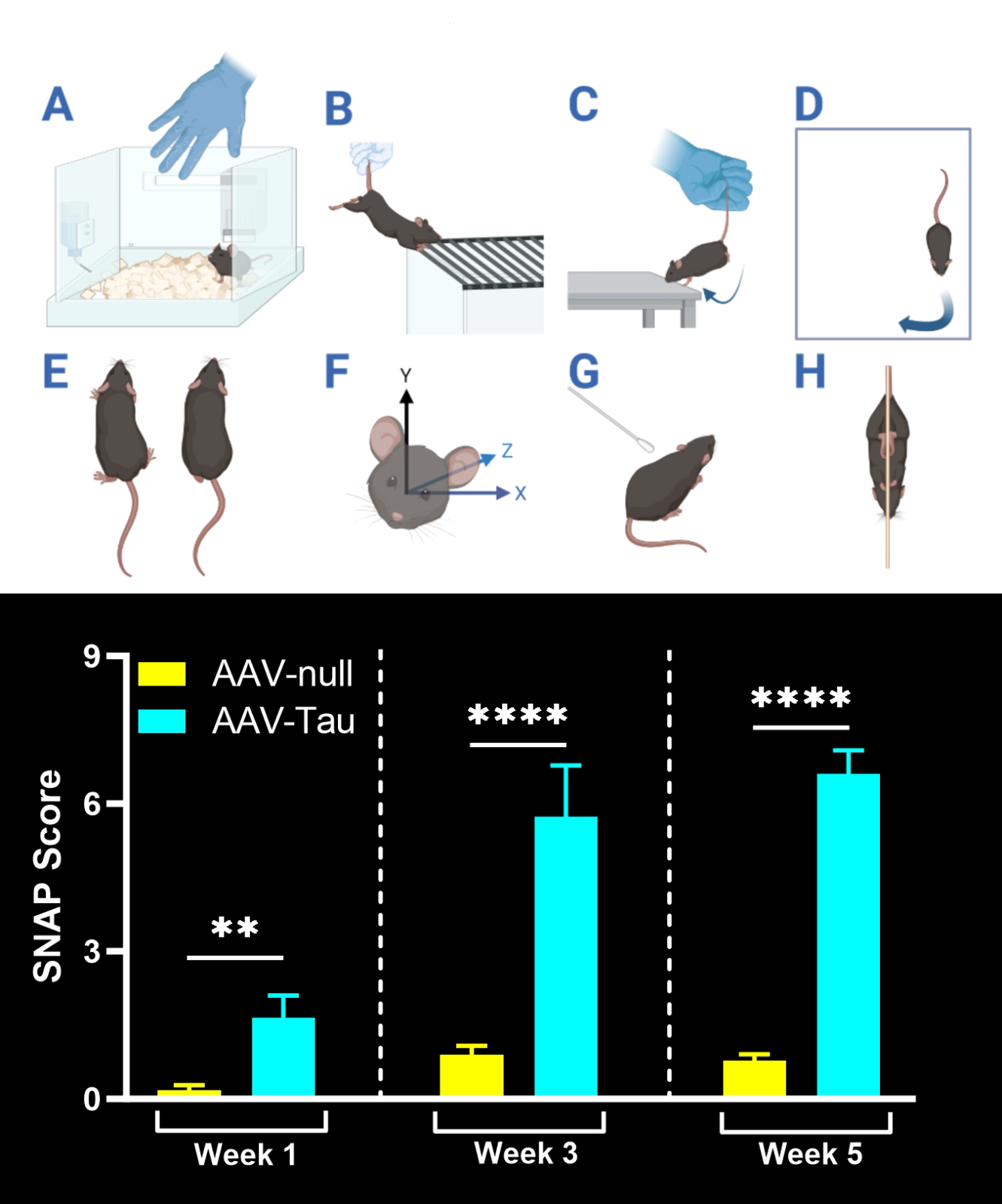
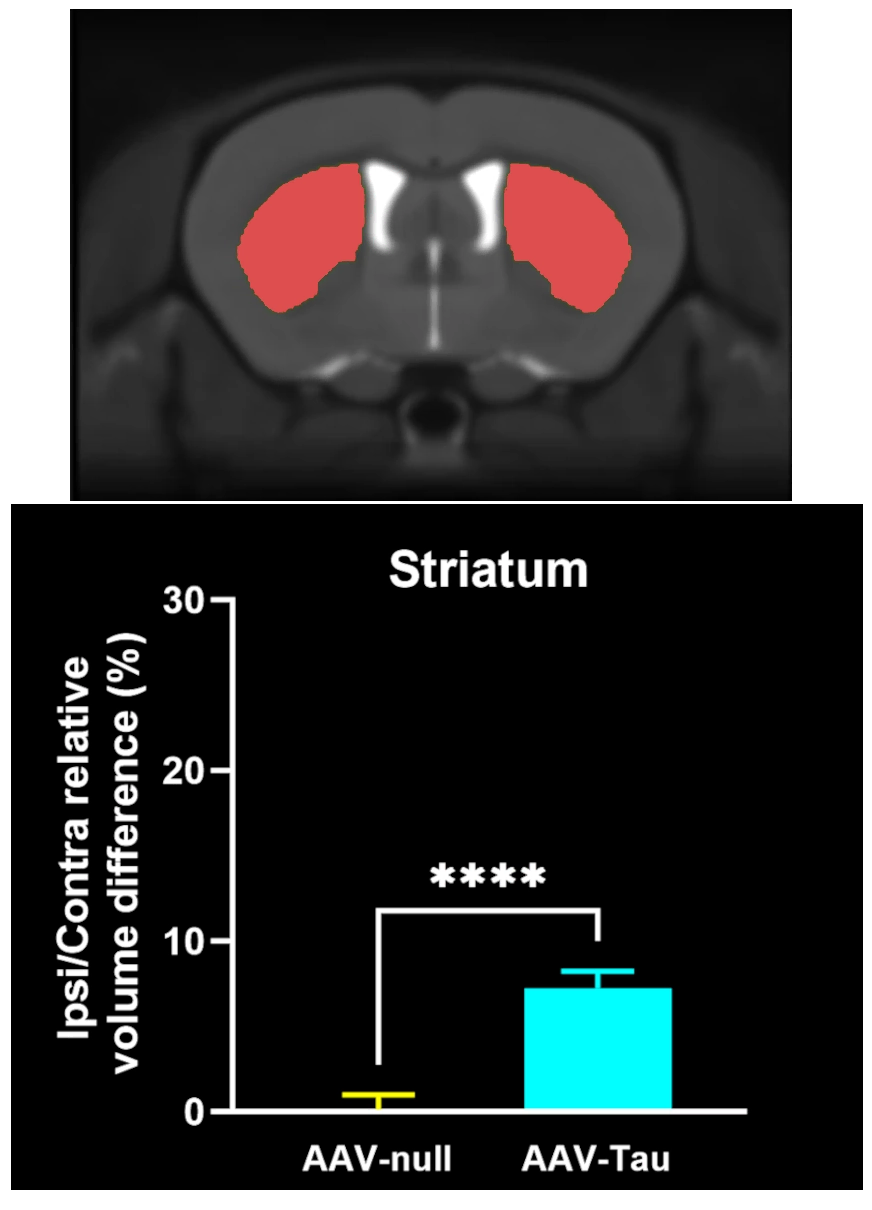
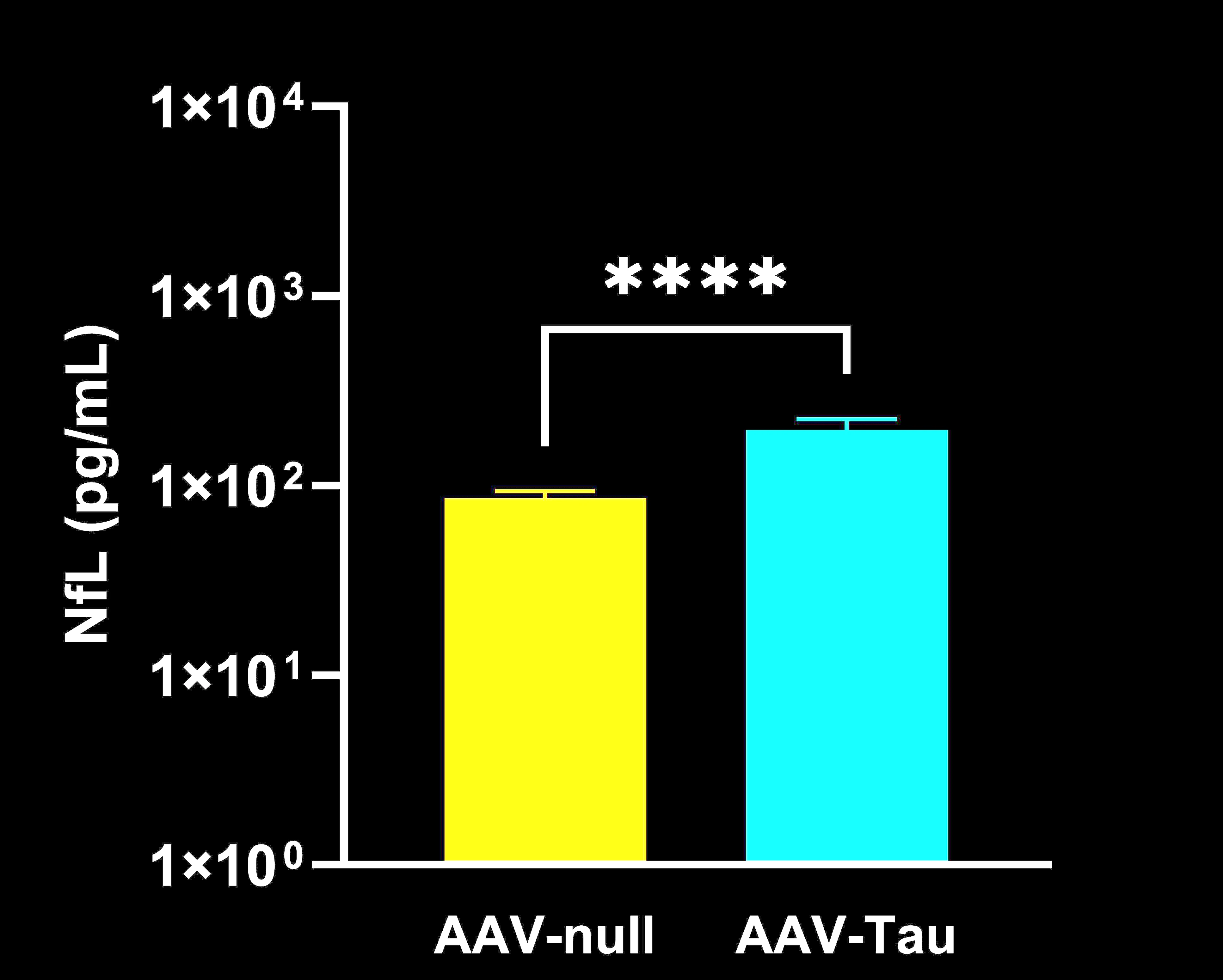
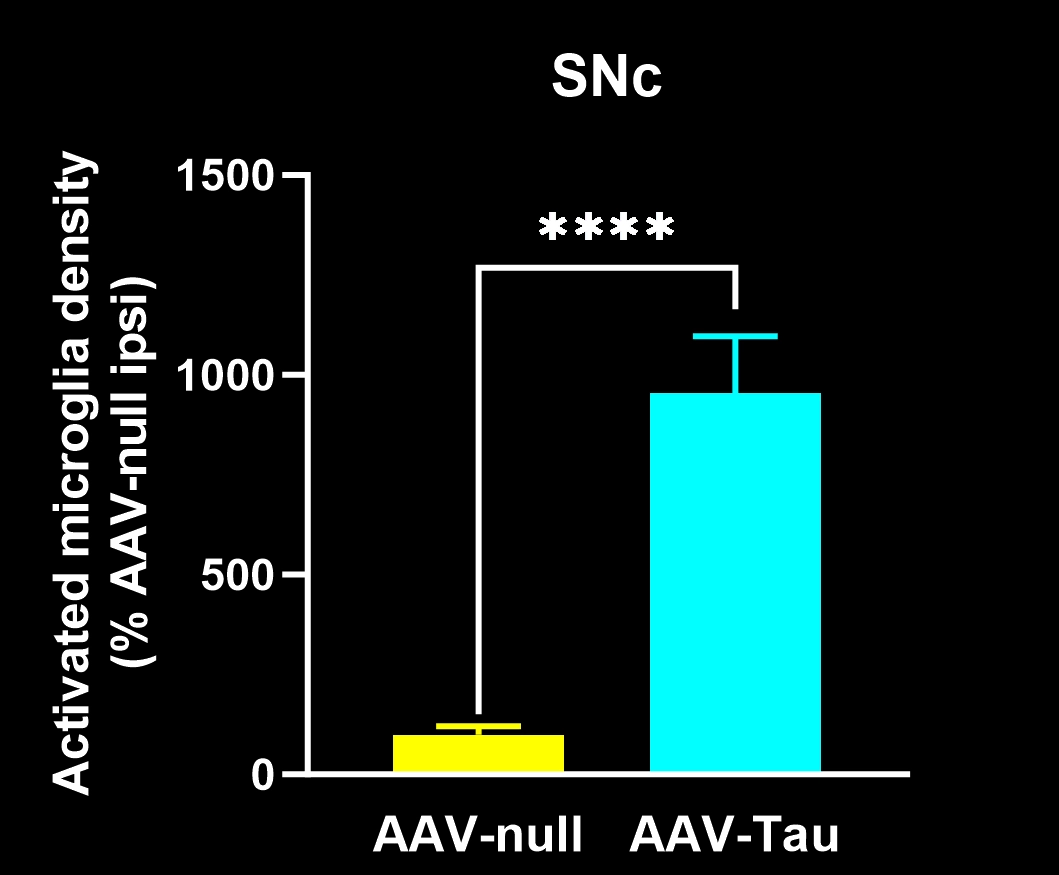
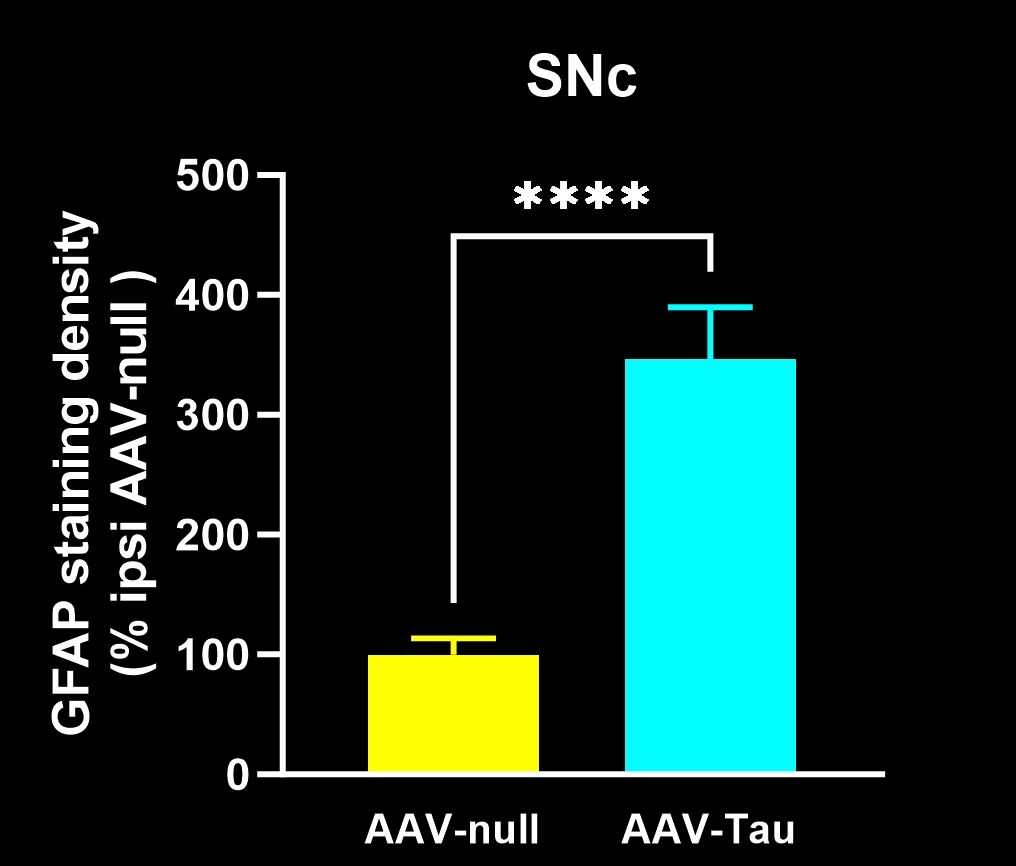
In our characterization of this model, we found:
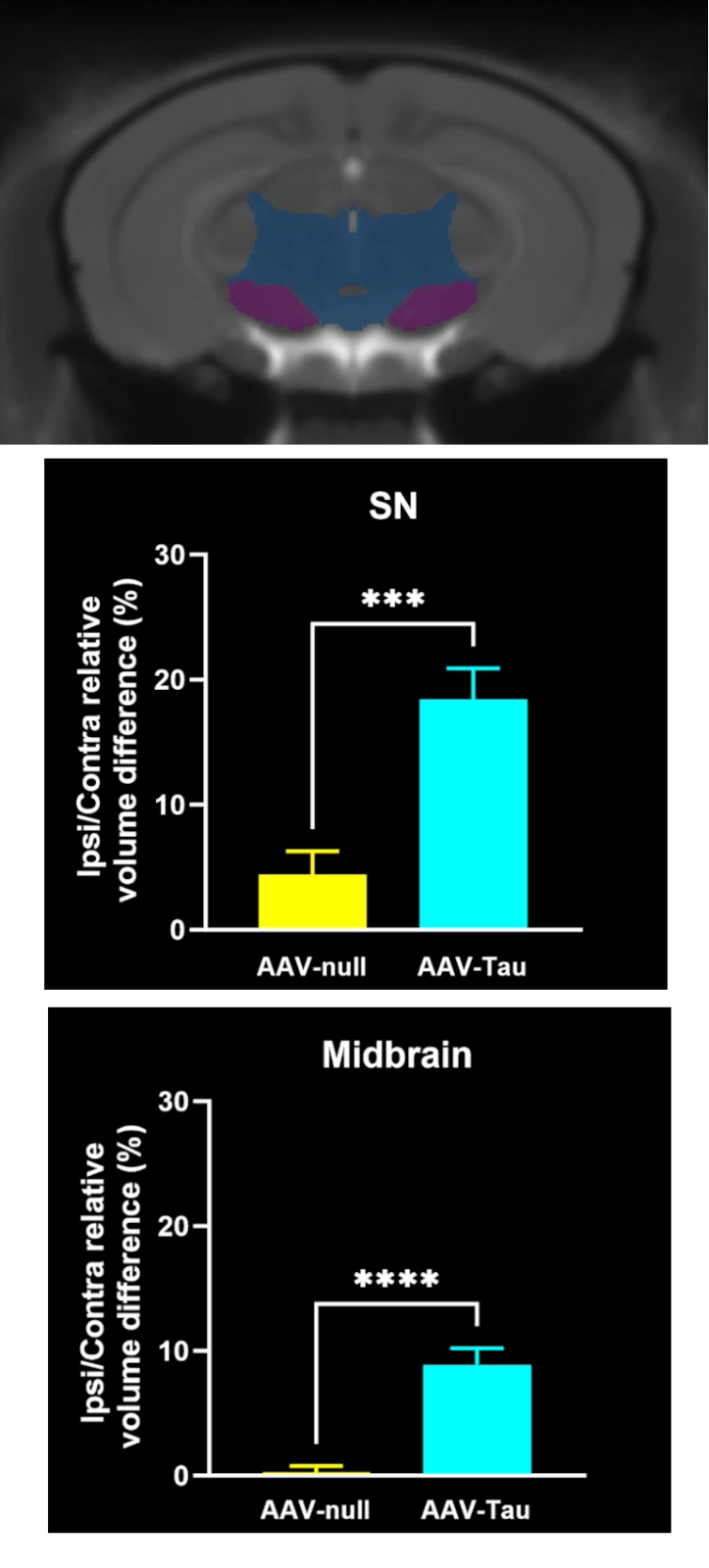
Phosphorylated tau aggregates in the substantia nigra and midbrain.
Activated microglia and reactive astrocytes in close proximity to the intracellular tau inclusions.
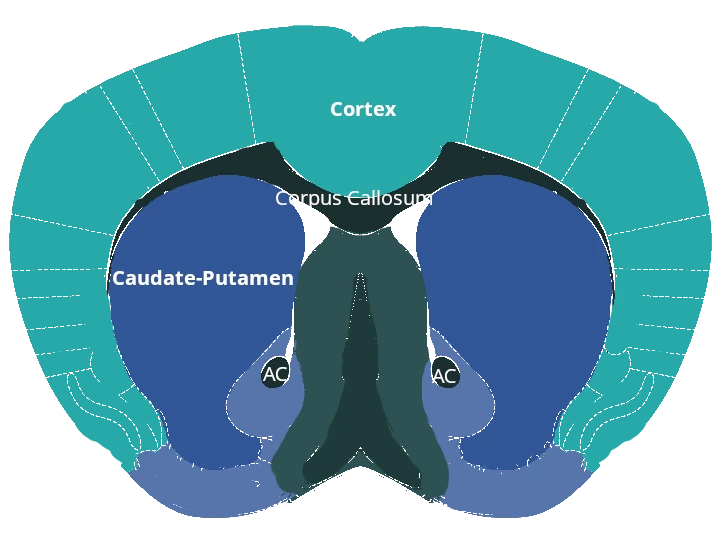
Loss of dopaminergic neurons in the substantia nigra pars compacta.
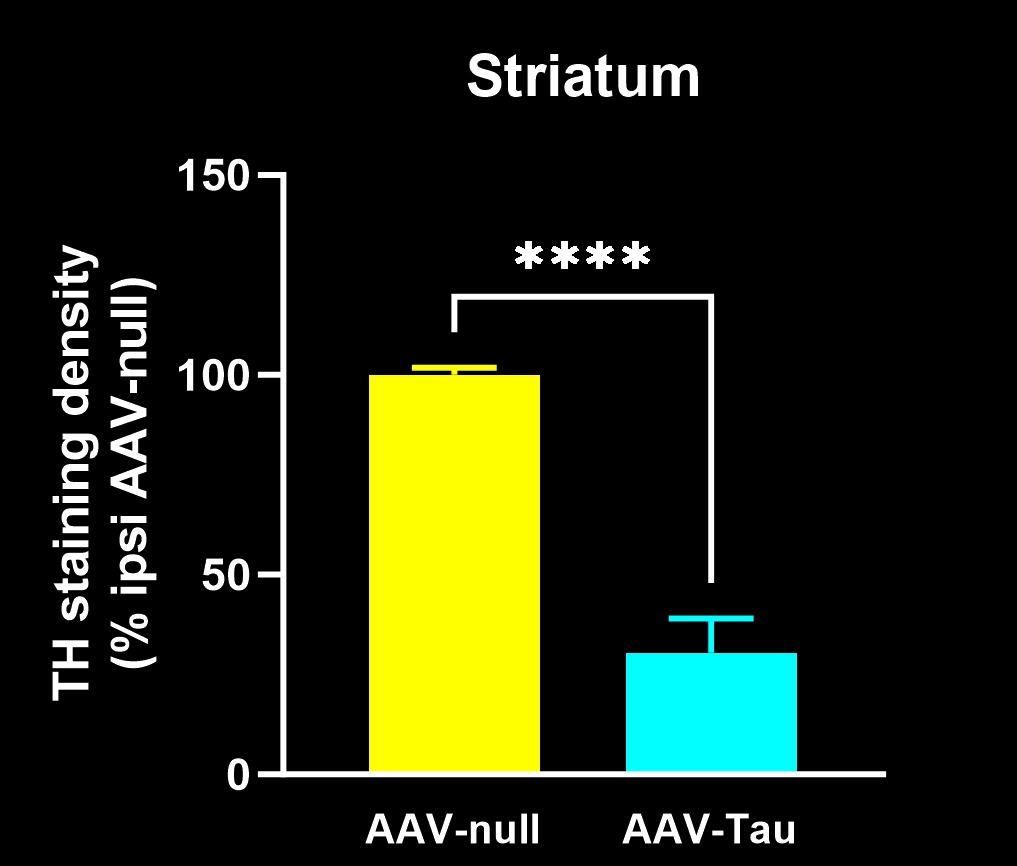
Reduction of dopaminergic terminals in the caudate-putamen.
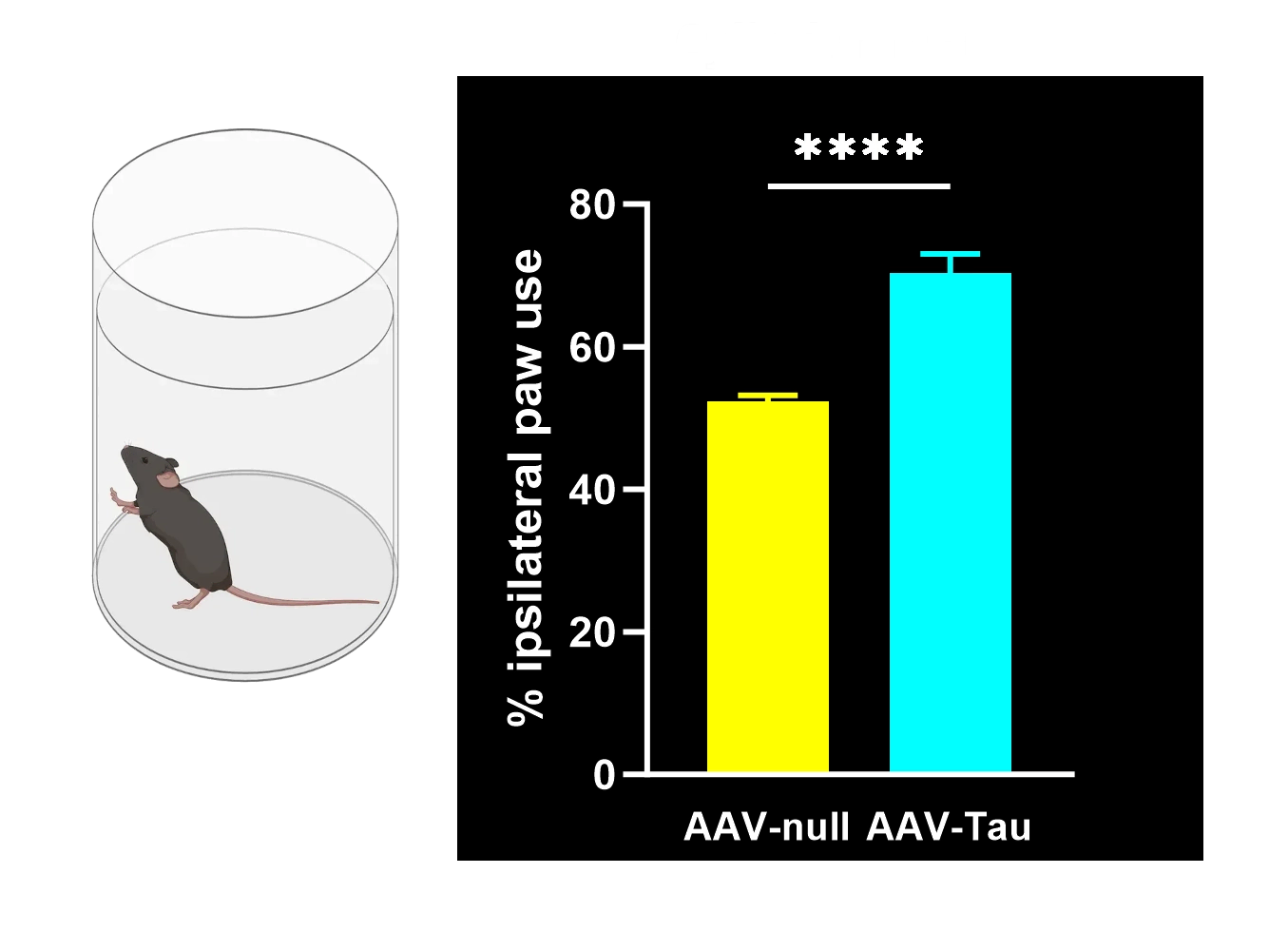
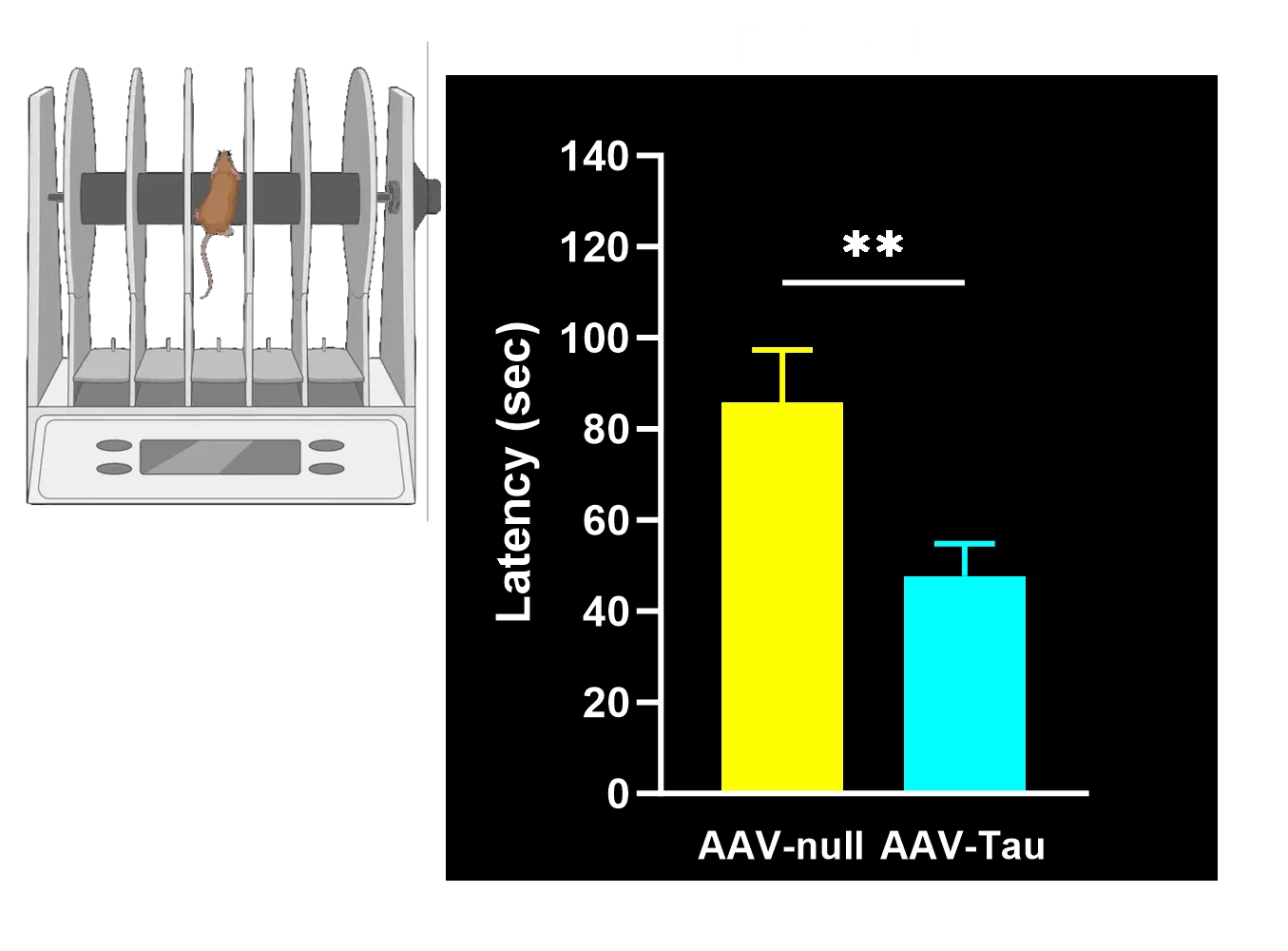
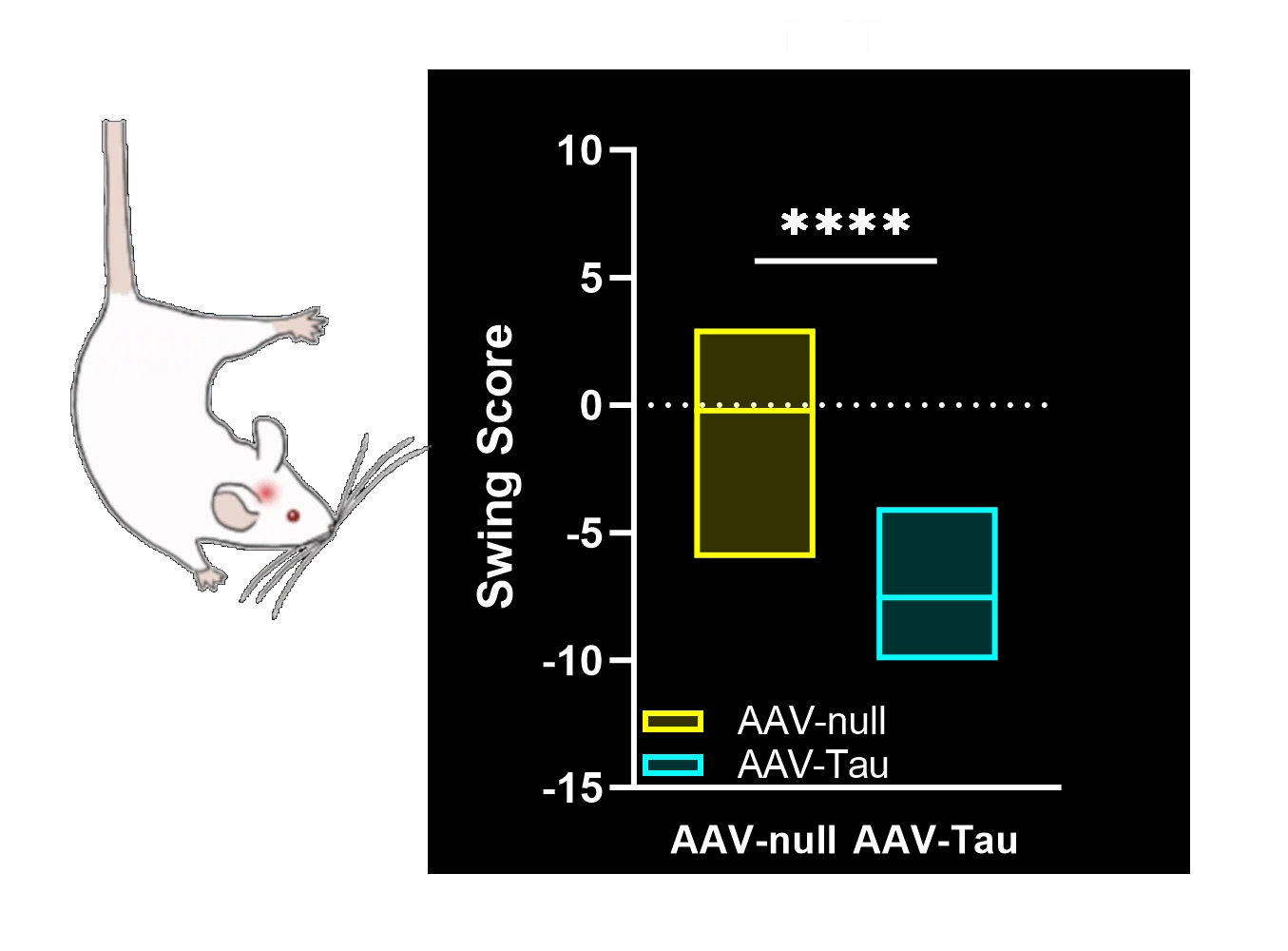
Motor deficits measured by hindlimb clasping, cylinder test, rotarod, tail suspension swing test, and SNAP scores.
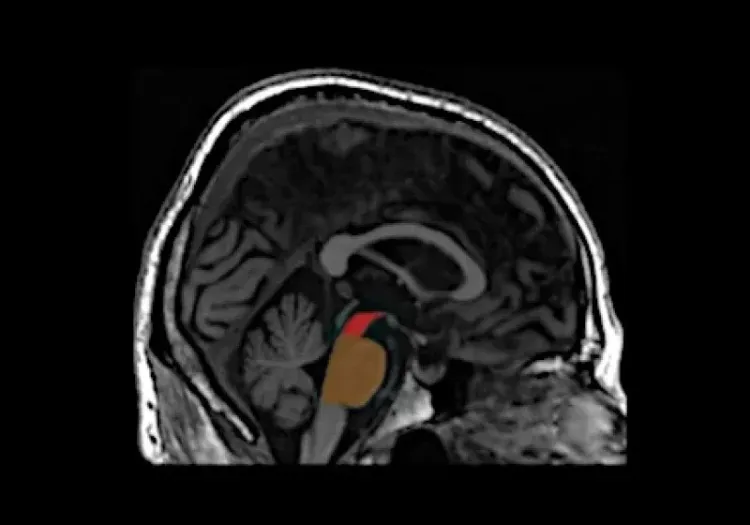
Regional MRI brain atrophy in the substantia nigra, midbrain, and striatum.
Elevated neurofilament light chain (NfL) concentrations in plasma.
Interactive Presentation of Our Research Study
In the “Image Interactive” below, you can find results from our comprehensive, multimodality characterization of our tauopathy mouse model, including high-resolution Multiplex Immunofluorescence brain tissue sections.
How to use Our Interactive Viewer
Navigate through the “Image Story” via the left-hand panel or the on-screen arrows. You can pan around high-resolution microscopy images with your mouse, and zoom in/out using the scroll wheel or the +/- controls. The Control Panel (top-right) allows toggling of image channels and segmentation overlays. For the best experience, we recommend switching to full-screen mode. This Interactive Presentation enables you to explore the model’s neuropathology and associated functional deficits in detail, as if looking directly down the microscope.
Image Interactive illustrating tau staining & analysis, including high-resolution Multiplex Immunofluorescence brain tissue sections, from our 4R tauopathy mouse model and control mice. This presentation highlights our tau IF staining & image analysis capabilities.
Key Advantages of Biospective's Tau Staining & Analysis Services:
High-sensitivity tau detection
Custom antibody/marker staining
High-throughput, automated whole slide imaging and neuroanatomical region analysis
Tau characterization and quantification
Glial cell morphology and phenotype analysis
Advanced neuroinflammation and Aβ plaque environment metrics — highly sensitive to small changes in disease progression
Cross-species (mouse, rat) compatibility
Complementary services (e.g. fluid biomarkers measured via immunoassays)
Contact Us CTA
To discuss your study requirements or request a quote for Tau Staining and Image Analysis services:
FAQs
FAQs
Keywords
Related Content
Up-to-date information on Microglia and Neuroinflammation.
PS19 Mouse Model for Tau Targeted Drug Development
An overview of PS19 (P301S) mice as a transgenic model for preclinical evaluation of disease-modifying therapies targeting tau seeding & spreading.
Microglia, Astrocytes & Tau in Neurodegenerative Diseases
How glial-driven neuroinflammation fuels tau aggregation, propagation, and neuronal loss in Alzheimer’s disease and other tauopathies.
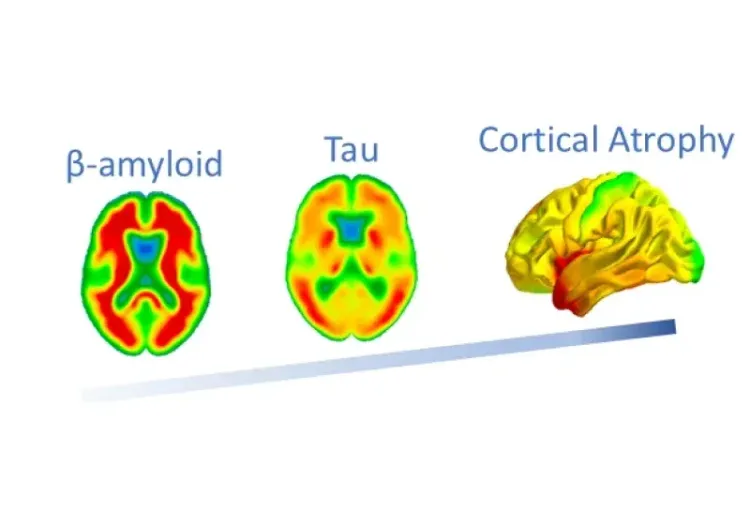
Tau-related Atrophy is Independent of β-Amyloid & APOE ε4
Using MRI, Tau PET, and Amyloid PET imaging biomarkers from the ADNI study, we show that Tau is more strongly linked to brain atrophy than β-amyloid or APOE ε4.
Decreased Brain Glucose Metabolism in MCI is Driven by Tau
We demonstrate a more significant role of tau than β-amyloid in the reduction of cerebral glucose metabolism by analysis of PET images from the ADNI study.
MRI Measures of Disease Progression for PSP Clinical Trials
MRI biomarkers (including brain atrophy) from the 4RTNI and FTLDNI natural history studies of Progressive Supranuclear Palsy (PSP).
MRI & Corticobasal Degeneration (CBD)
Longitudinal MRI brain atrophy measures from the 4RTNI and FTLDNI studies including sample size calculations for clinical trials of Corticobasal Degeneration.
Microglia-Neuron Interactions & Neurodegenerative Diseases
A concise review of the direct interactions between microglia & neurons, and how these cell-to-cell interactions may be affected in neurodegenerative diseases.
Amyloid-β & Inflammatory Microenvironment in Alzheimer's Mice
We have analyzed the complex spatial relationships between β-amyloid plaques, activated & resting microglia, and astrocytes in an APP/PS1 transgenic model.
Microglial Activation in an α-Synuclein PFF Mouse Model
We have quantified microglial activation, based on morphology, in an α-synuclein preformed fibril (PFF) seeding & spreading mouse model of Parkinson’s disease.
Microglia Morphology in ALS, Alzheimer's Disease & Parkinson's Disease
An overview of microglial morphological analysis and the applications to neurodegenerative disease research and drug discovery & development.
Mitochondrial Dysfunction in Microglia & Astrocytes
The role of mitochondrial dysfunction in microglia and astrocytes in neurodegenerative diseases, including Alzheimer’s disease, Parkinson’s disease, and ALS.
Lysosome Dysfunction in Microglia & Astrocytes
An overview of lysosomal dysfunction in microglia & astrocytes, and its role in neurodegenerative diseases.
NLRP3 Inflammasome and Neurodegenerative Diseases
An overview of the NLRP3 inflammasome and its role in neurodegenerative diseases, including Alzheimer's disease, Parkinson’s disease, and ALS.
Impaired Microglia Autophagy in Neurodegenerative Diseases
How impaired microglia autophagy contributes to the progression of neurodegenerative diseases.
TNF-α (TNF-alpha) & Microglia in Neurodegenerative Diseases
An overview of the function of tumor necrosis factor-alpha (TNF-α) in microglia and its contribution to the progression of neurodegeneration.