Characterization of a Novel AAV-hTau Mouse Model of Tauopathies with Parkinsonian Features
Biospective's tau mouse model of PSP & CBD is a gene delivery–based preclinical model that induces rapid tau aggregation, dopaminergic neurodegeneration, neuroinflammation, and motor deficits. This unique mouse model of primary 4R tauopathy with Parkinsonian features was developed and characterized by Biospective to fill the need for a robust model of PSP & CBD that is well-suited for drug development studies.
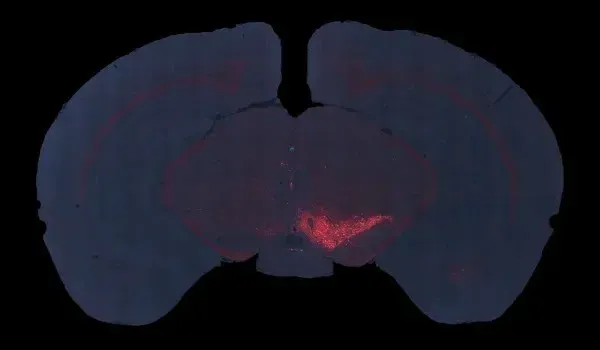
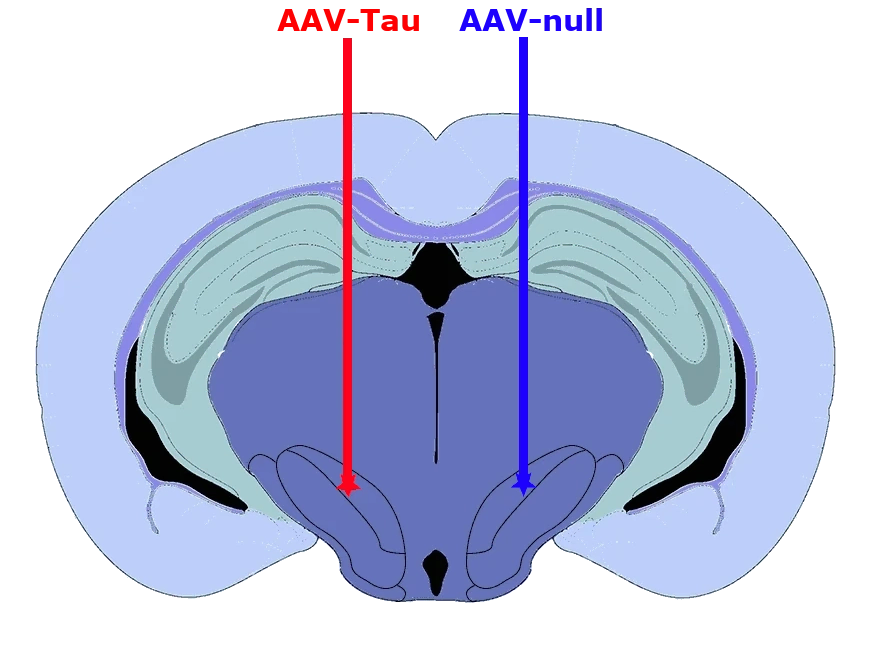
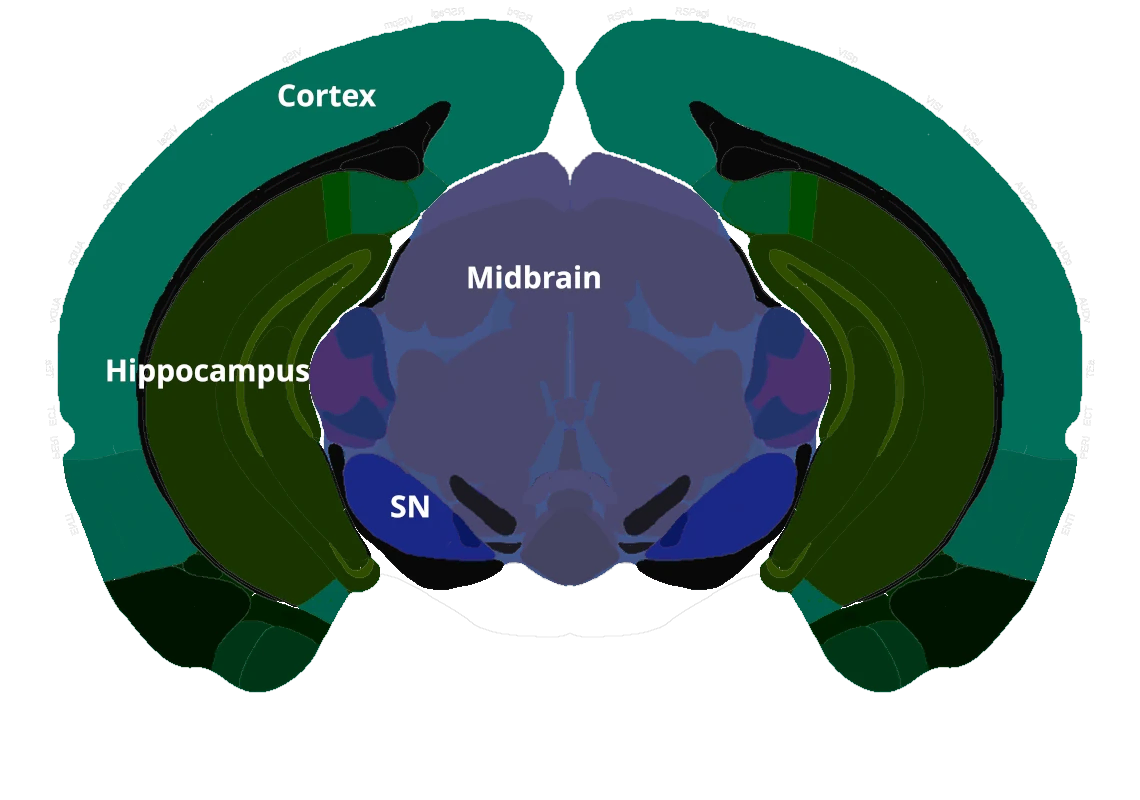
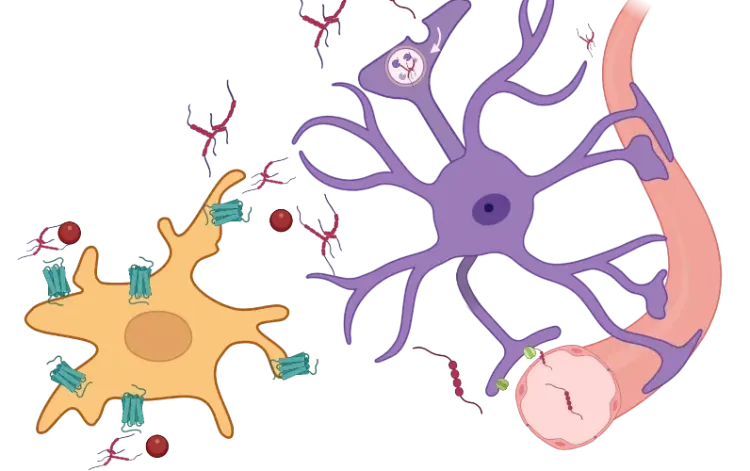
In this robust mouse model, adeno-associated virus (AAV) vectors expressing human wild-type tau (2N4R) are delivered into the substantia nigra pars compacta via stereotaxic injection. The resulting model demonstrates tau aggregates, dopaminergic neuron loss, neuroinflammation (activated microglia and astrocytes), and motor deficits reminiscent of human 4-repeat (4R) tauopathies (PSP and CBD).
Biospective has extensively characterized this animal model and leverages it as an ideal platform for preclinical drug development, supporting high-throughput efficacy testing, mechanism-of-action studies, and target engagement evaluation for novel therapeutics for tauopathies. As a specialized neuroscience CRO, Biospective provides fully integrated, end-to-end services – from surgical model induction and in vivo imaging to translational fluid biomarker assays and quantitative pathology – delivering high-quality data for its biotech and pharma sponsors worldwide.
Overview of Our Tau Mouse Model of PSP & CBD
A rapidly inducible tau animal model of 2N4R tauopathies optimized for preclinical drug development.
In this model, adeno-associated virus (AAV) vectors encoding human wild-type 2N4R tau undergo stereotaxic injection into the midbrain substantia nigra of young adult C57BL/6 mice. This targeted intra-nigral delivery drives high levels of toxic tau expression, triggering a cascade of Parkinsonian-like pathology. Our AAV-Tau model faithfully recapitulates key hallmarks of human PSP & CBD, including:
-
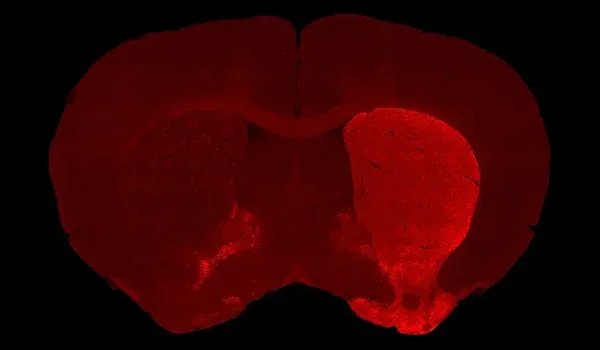
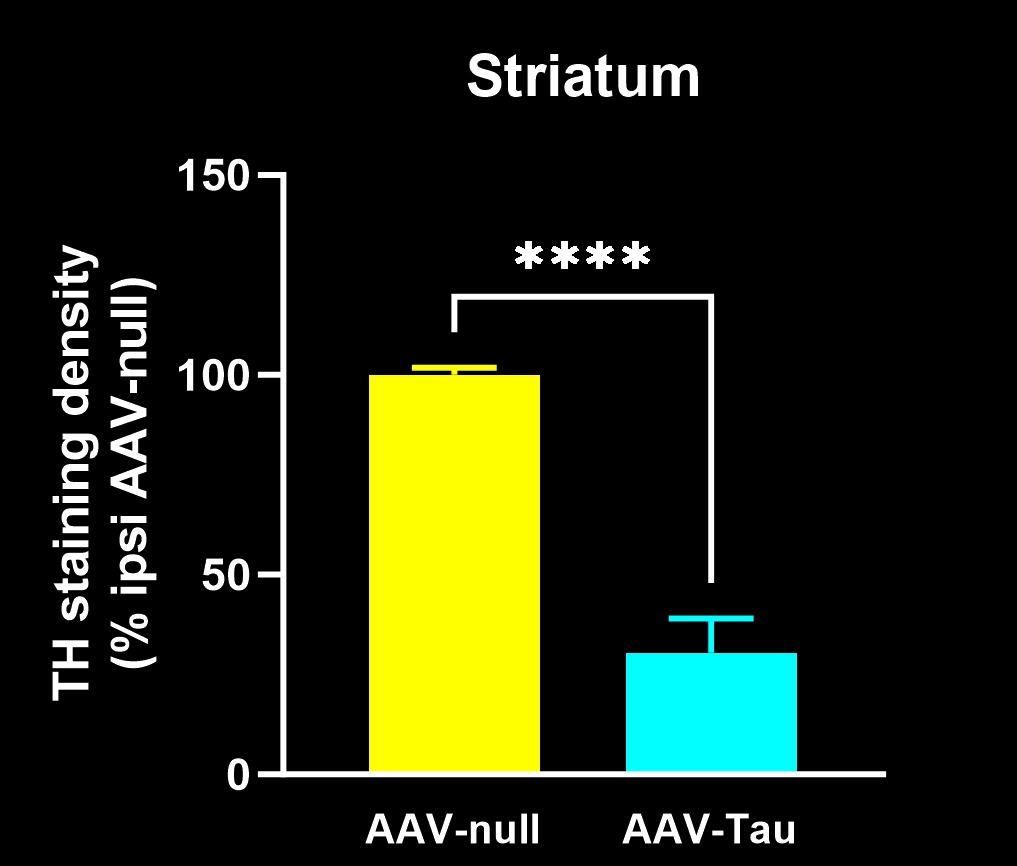
Dopaminergic neuron loss: Substantial degeneration of dopamine-producing neurons in the substantia nigra pars compacta, with corresponding loss of dopaminergic nerve fibers in the striatum.
-
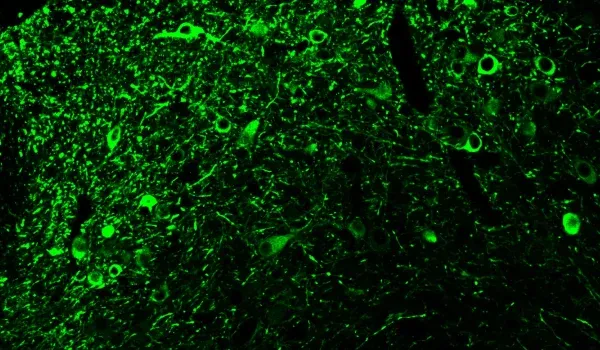
Tau aggregation: Accumulation of pathogenic human tau (especially phosphorylated tau), forming neurofibrillary tangle–like intracellular inclusions in affected brain regions.
-
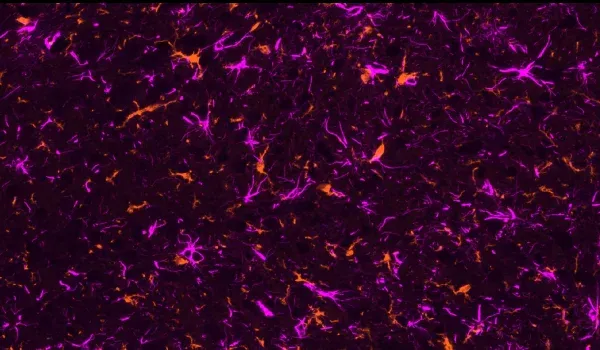
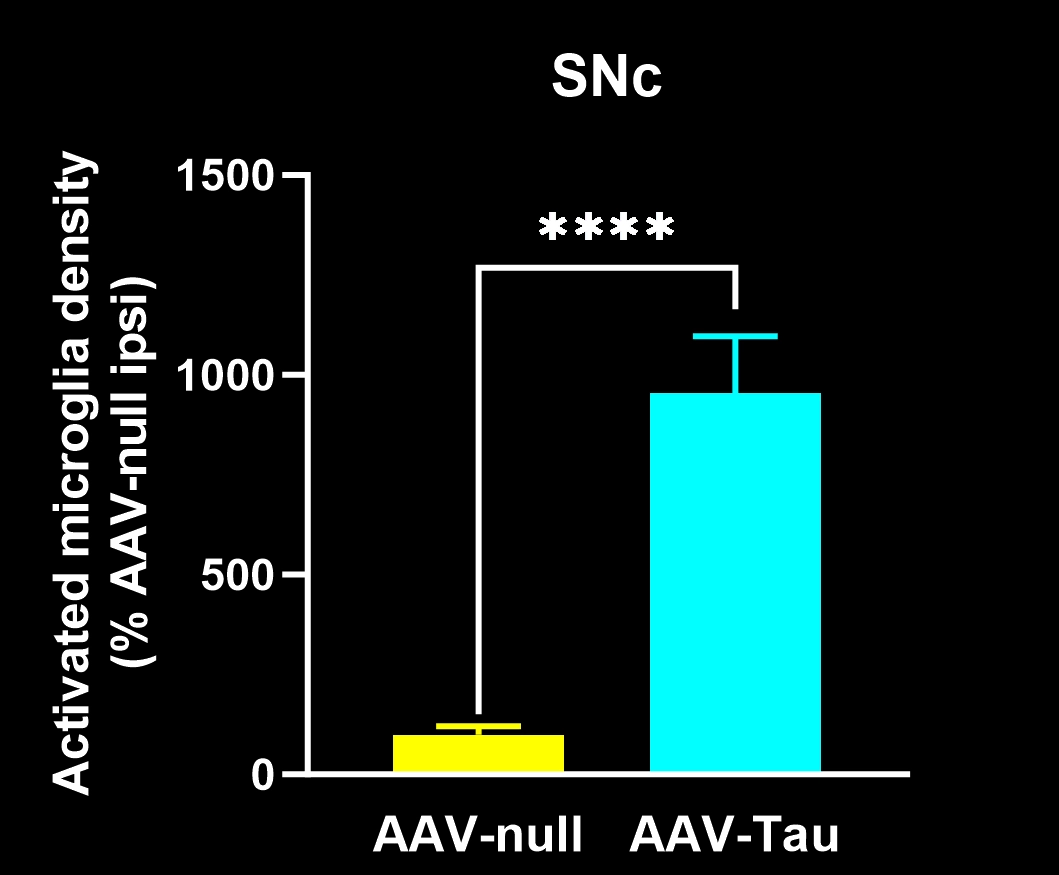
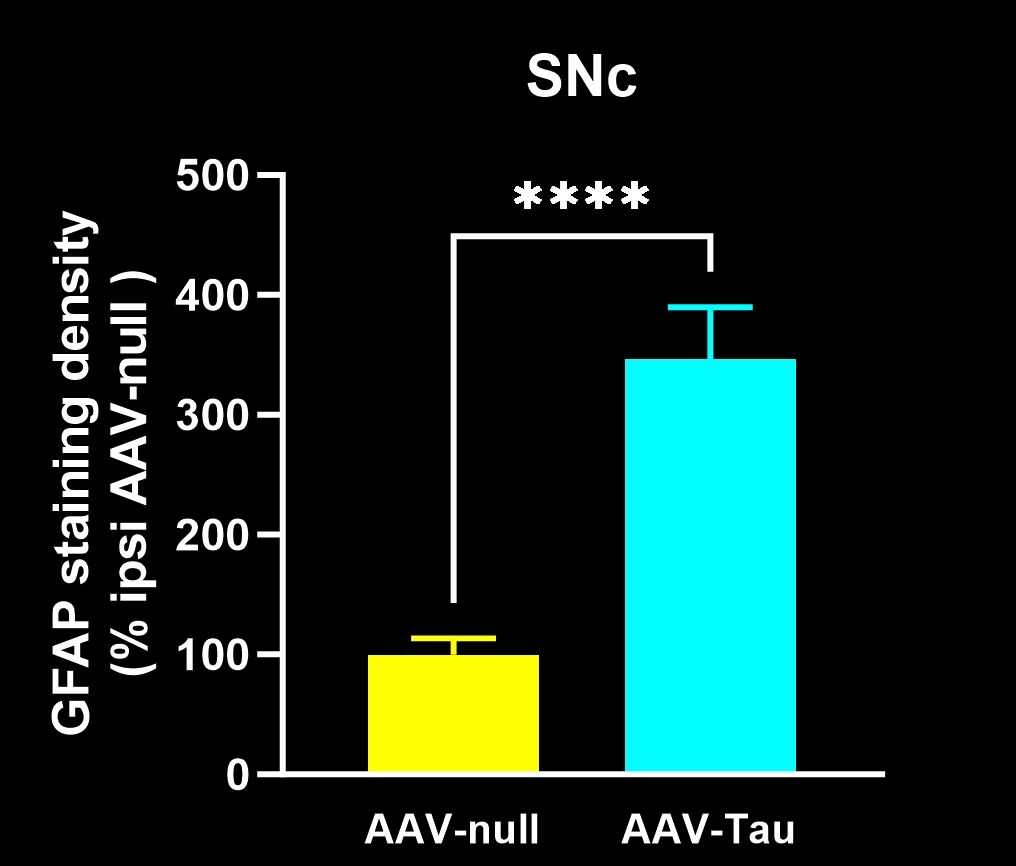
Robust neuroinflammation: Pronounced activation of microglia and reactive astrocytes in areas of tau pathology, mirroring the neuroinflammatory response observed in human tauopathies.
-
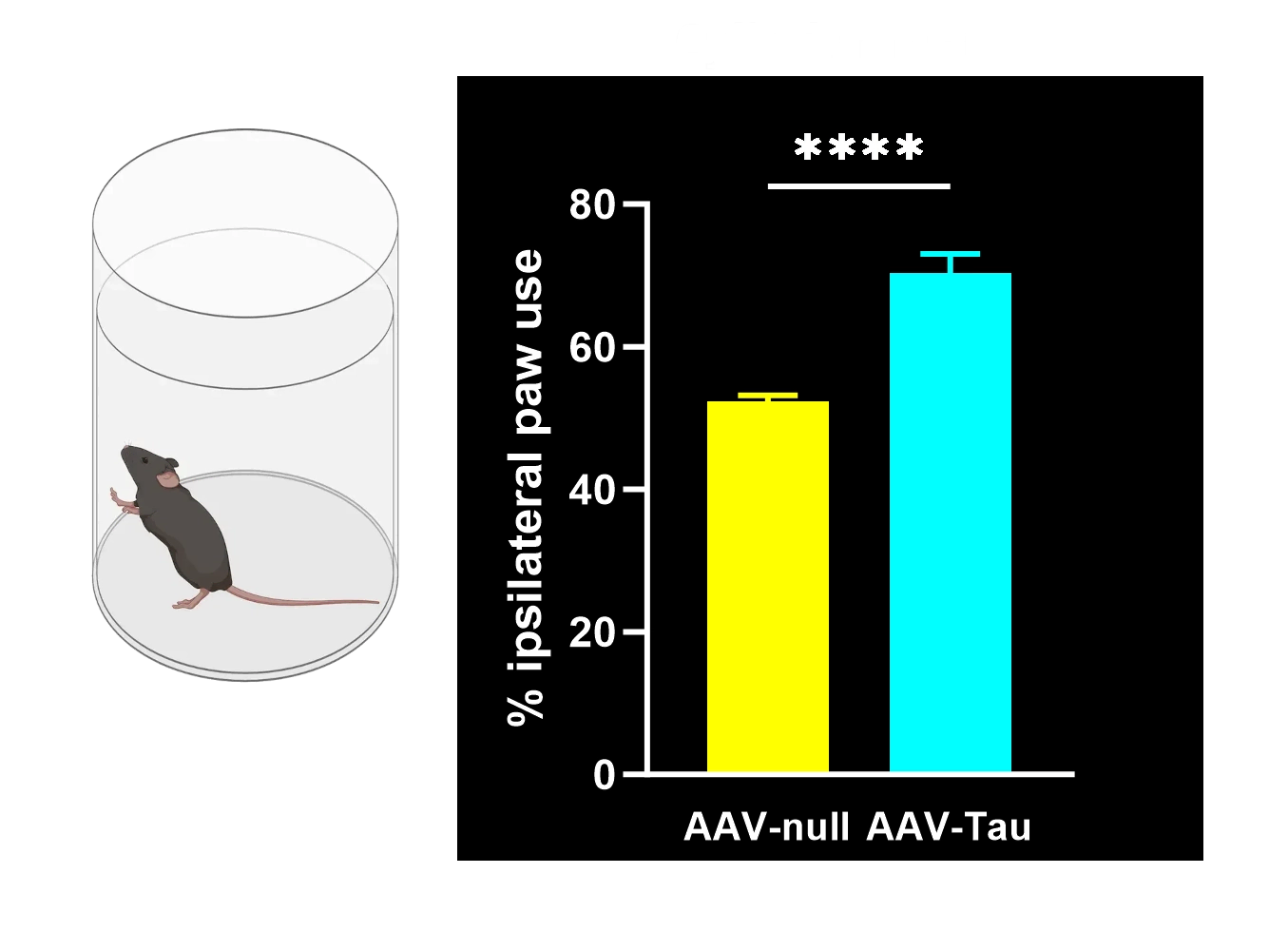
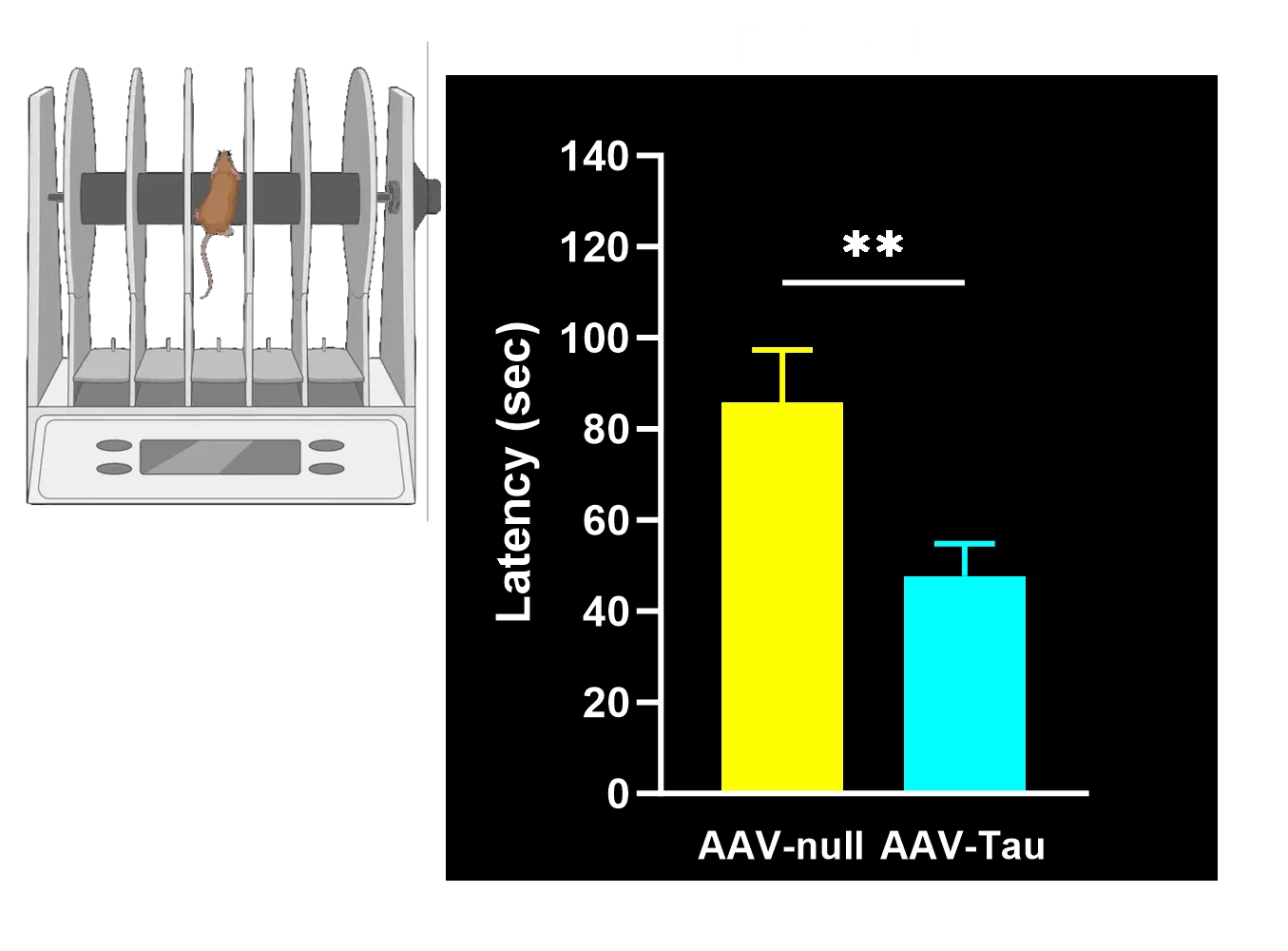
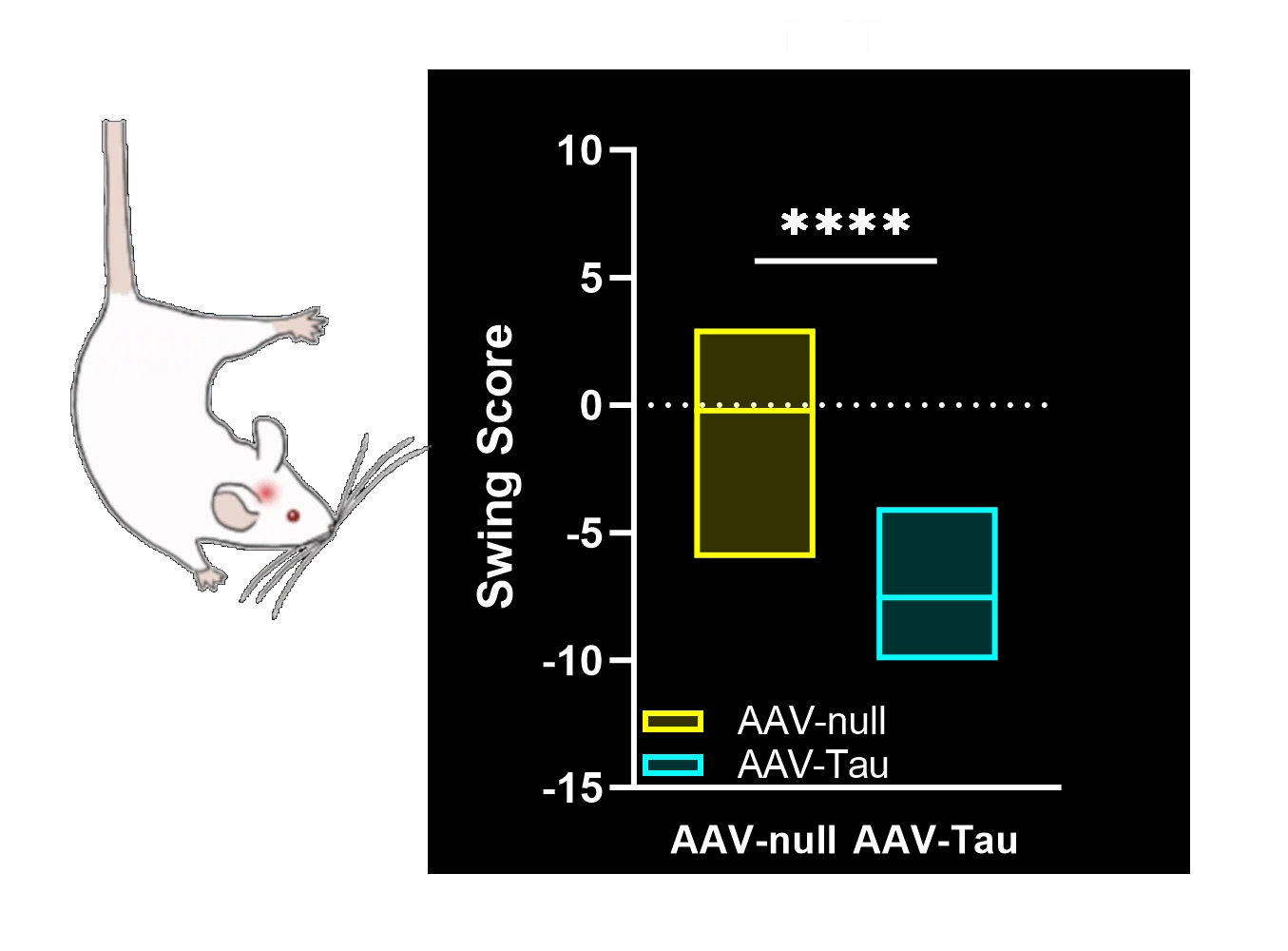
Motor function deficits: Significant motor impairments (e.g. limb use asymmetry, coordination and balance deficits) arising from nigrostriatal neurodegeneration, reminiscent of Parkinsonian motor symptoms.
By reproducing these pathological features, our AAV-Tau model provides a disease-relevant platform to evaluate therapeutic interventions under conditions that mirror the clinical hallmarks of human 2N4R tauopathies.
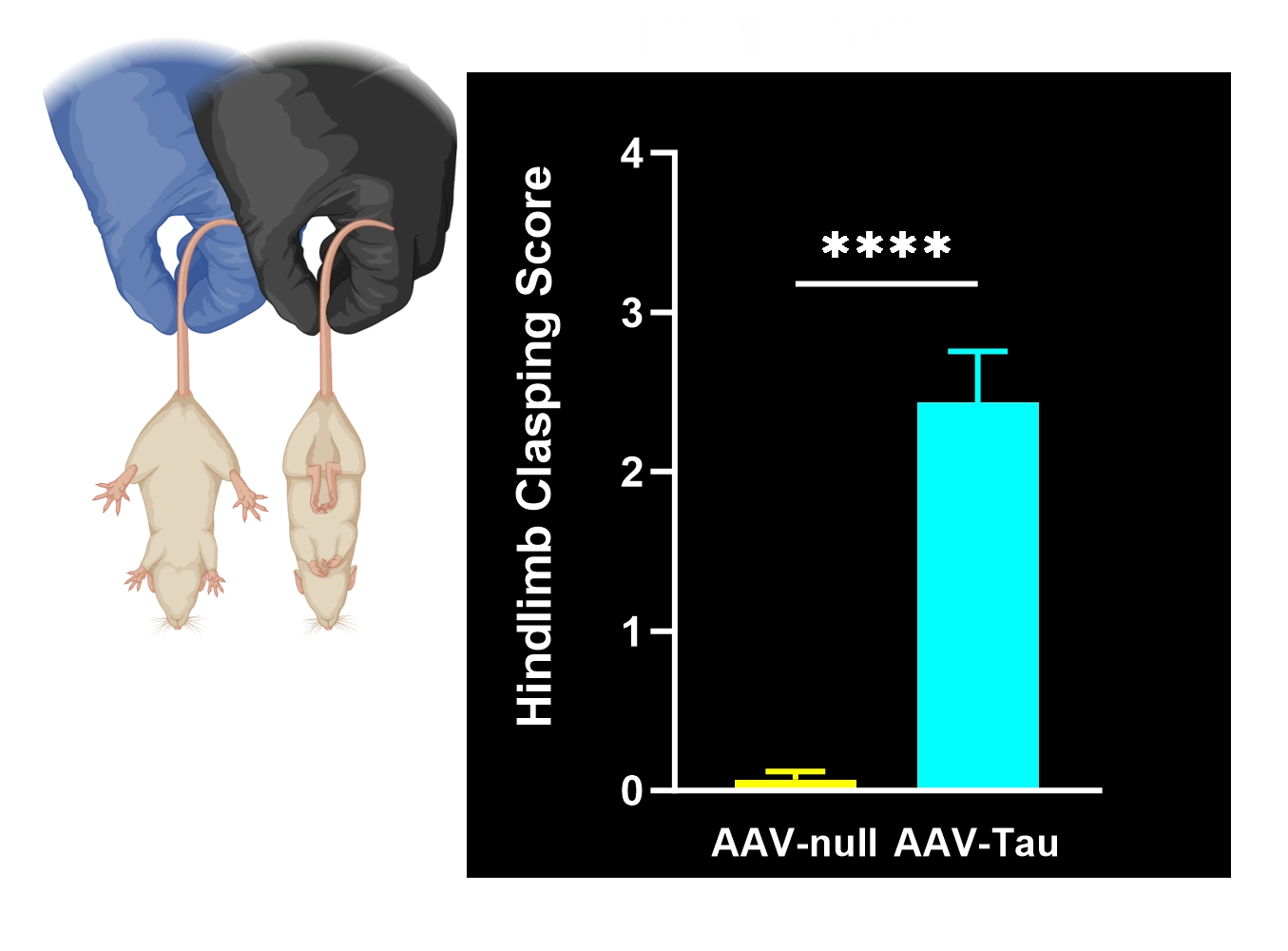
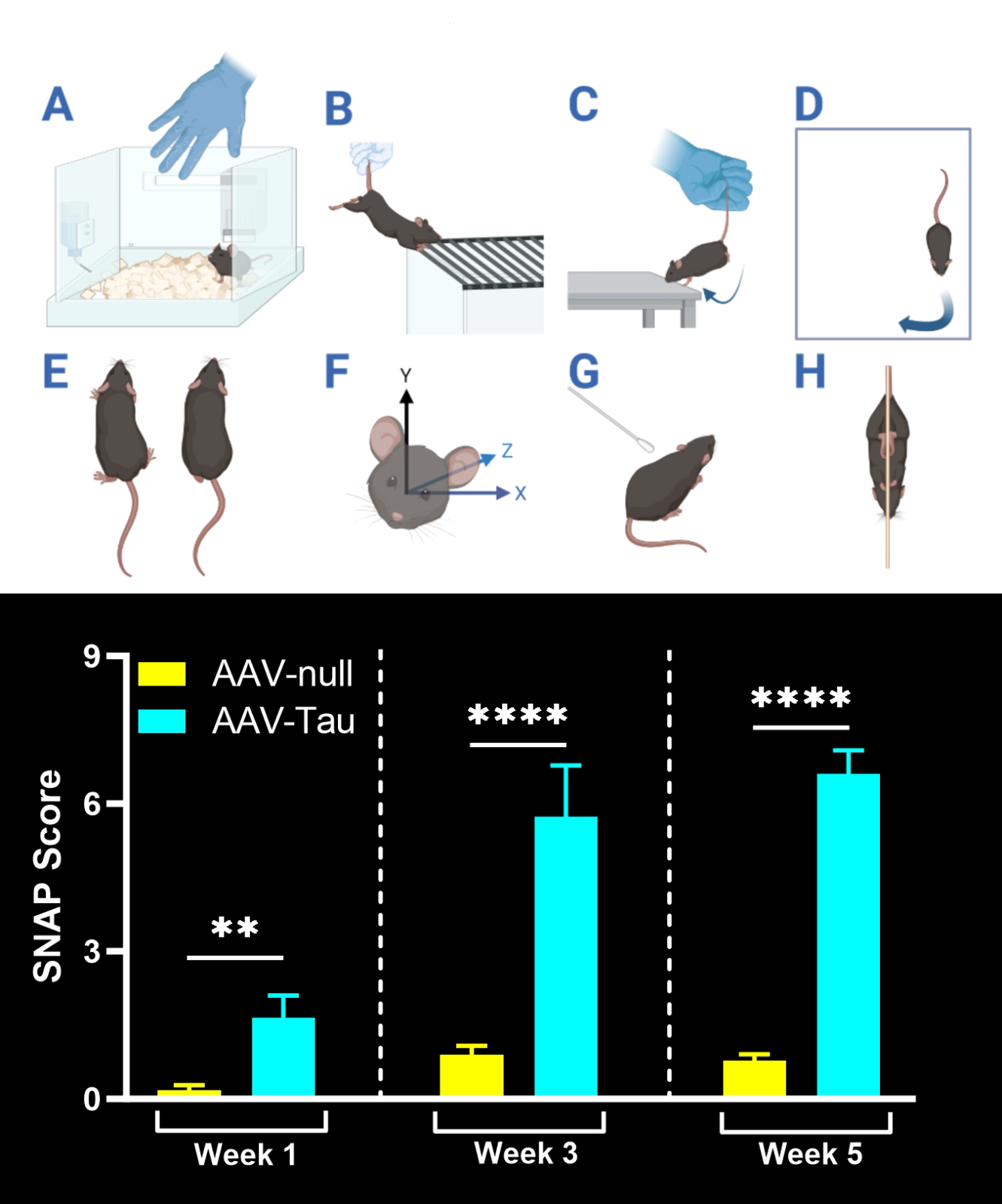
Given the lack of dedicated mouse models for PSP and CBD that show Parkinsonian motor dysfunction characteristic of human disease, our proprietary model fills this gap. Our AAV-Tau model yields rapid and robust disease manifestation on an accelerated timeline. These mice develop measurable neuron loss and motor deficits within weeks of vector injection. Typically, by ~6 weeks post-injection, significant dopaminergic neuron loss in the substantia nigra is observed alongside clear behavioral deficits. This fast onset enables shorter studies and faster go/no-go decisions in preclinical programs without sacrificing biological relevance. The compressed in vivo timeline makes the AAV-Tau model highly suited for high-throughput efficacy screening and proof-of-concept studies in tauopathy research.
AAV-hTau Model Generation & Study Timeline
Our expert team employs state-of-the-art, precise stereotaxic surgery techniques to generate the AAV-hTau model with minimal study start-up times.
We inject high-titer AAV vectors unilaterally into the midbrain substantia nigra (SN) region of ~12-week-old C57BL/6 mice. In these surgeries, we utilize digital stereotaxic systems with automated microinjectors to ensure accurate targeting and controlled viral delivery. This refined methodology yields consistent tau expression in the substantia nigra and midbrain.
Following injection, Parkinsonian neuropathology evolves rapidly. Animals begin to exhibit motor deficits (e.g. limb use asymmetry) within a few weeks post-injection, coinciding with ongoing dopaminergic neuron loss and protein aggregation in the affected brain regions. By ~6 weeks post-AAV injection, robust disease endpoints can be captured.
An illustration of the process for AAV-hTau mouse model generation.
This abbreviated timeline is a significant advantage as it enables quicker iteration and rapid data readouts, in contrast to transgenic models that might require many months to develop endpoints. The fast onset and severity of pathology in our AAV-hTau model, therefore, provide an efficient system for testing therapeutic efficacy and mechanistic hypotheses in preclinical tau programs. Biospective can initiate studies with this model on-demand, thanks to our in-house capabilities (including ready access to viral vector stock), ensuring minimal startup time for your project.
Validated Endpoints & Translational Biomarkers
Biospective has implemented a suite of validated endpoints and tauopathy relevant biomarkers to enable clinical advancement of therapeutic programs.
To fully characterize the AAV-hTau model and assess treatment outcomes, Biospective has validated a broad spectrum of endpoints – encompassing behavioral assays, neuroimaging, fluid biomarkers, and histopathology. This comprehensive approach yields robust, quantitative readouts for both efficacy and mechanism-of-action in preclinical studies. Key validated endpoints in our tauopathy model include:
Behavioral & Functional Endpoints
-
Hindlimb Clasping Test: A sensitive indicator of neurodegeneration (brainstem/spinal reflex integrity) often observed as disease progresses.
-
Tail Suspension Swing Test: Assesses lateral bias in movement during a tail suspension, indicating unilateral motor deficits resulting from nigrostriatal damage.
-
Cylinder Test: Measures forelimb use asymmetry during rearing and exploratory behavior. Reduced use of the contralateral paw reflects motor impairment on the side of the lesion.
Imaging, Fluid & Tissue Biomarkers
-
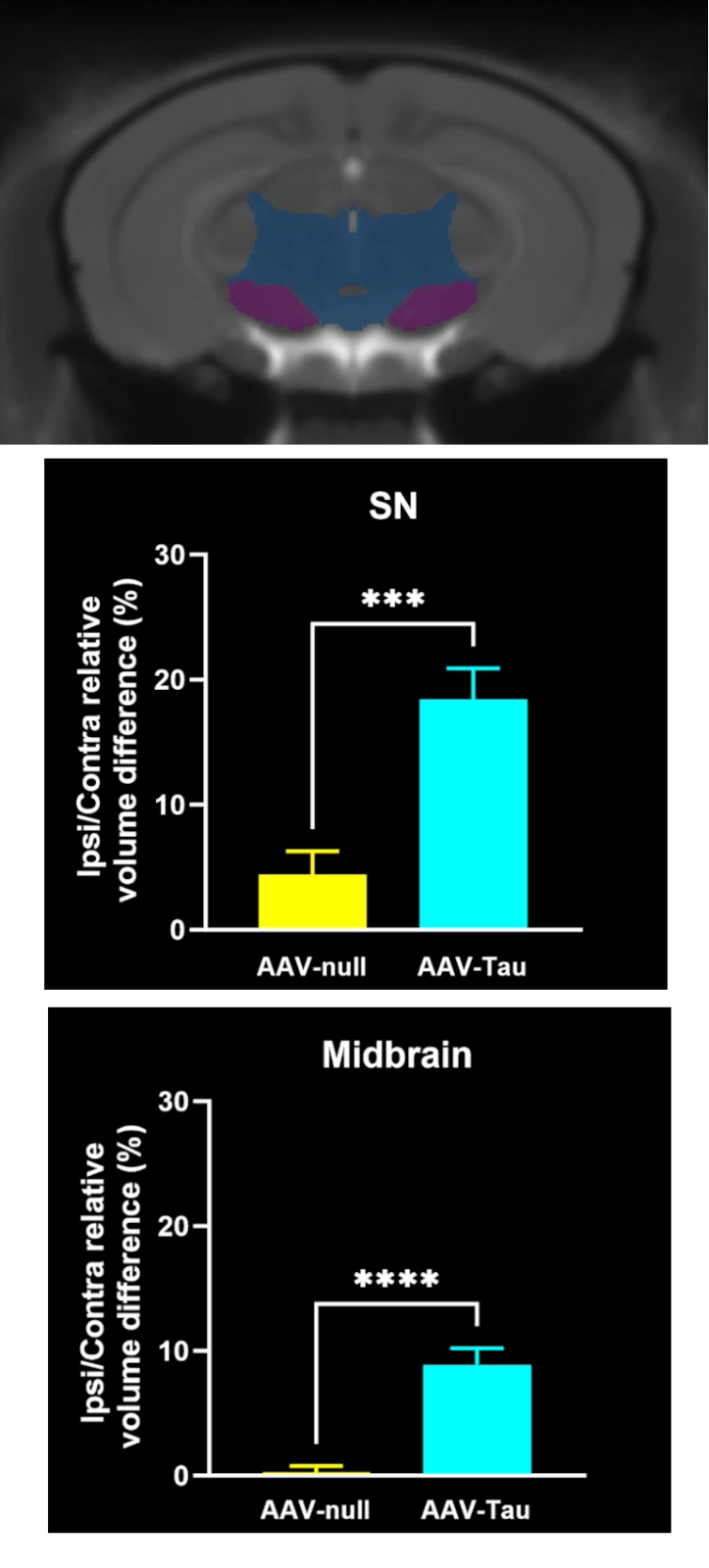
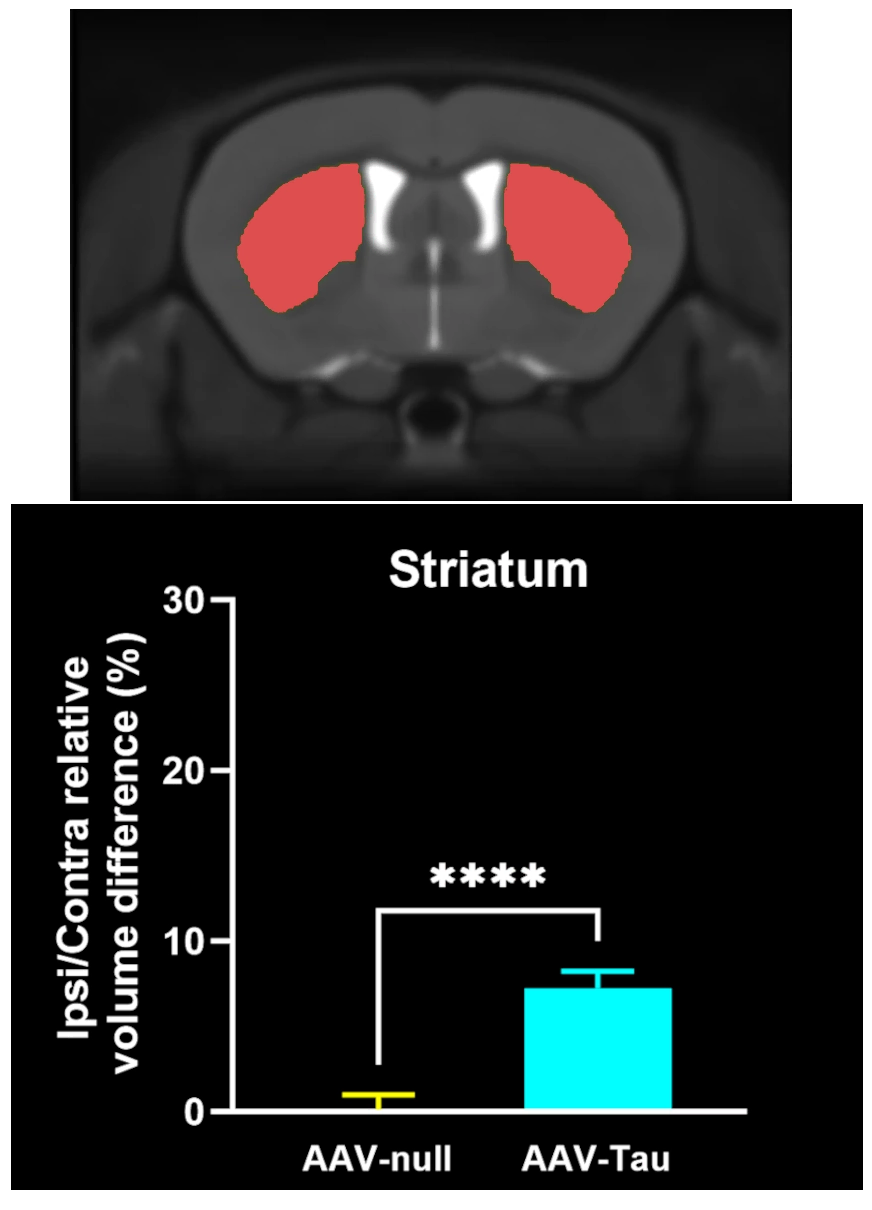
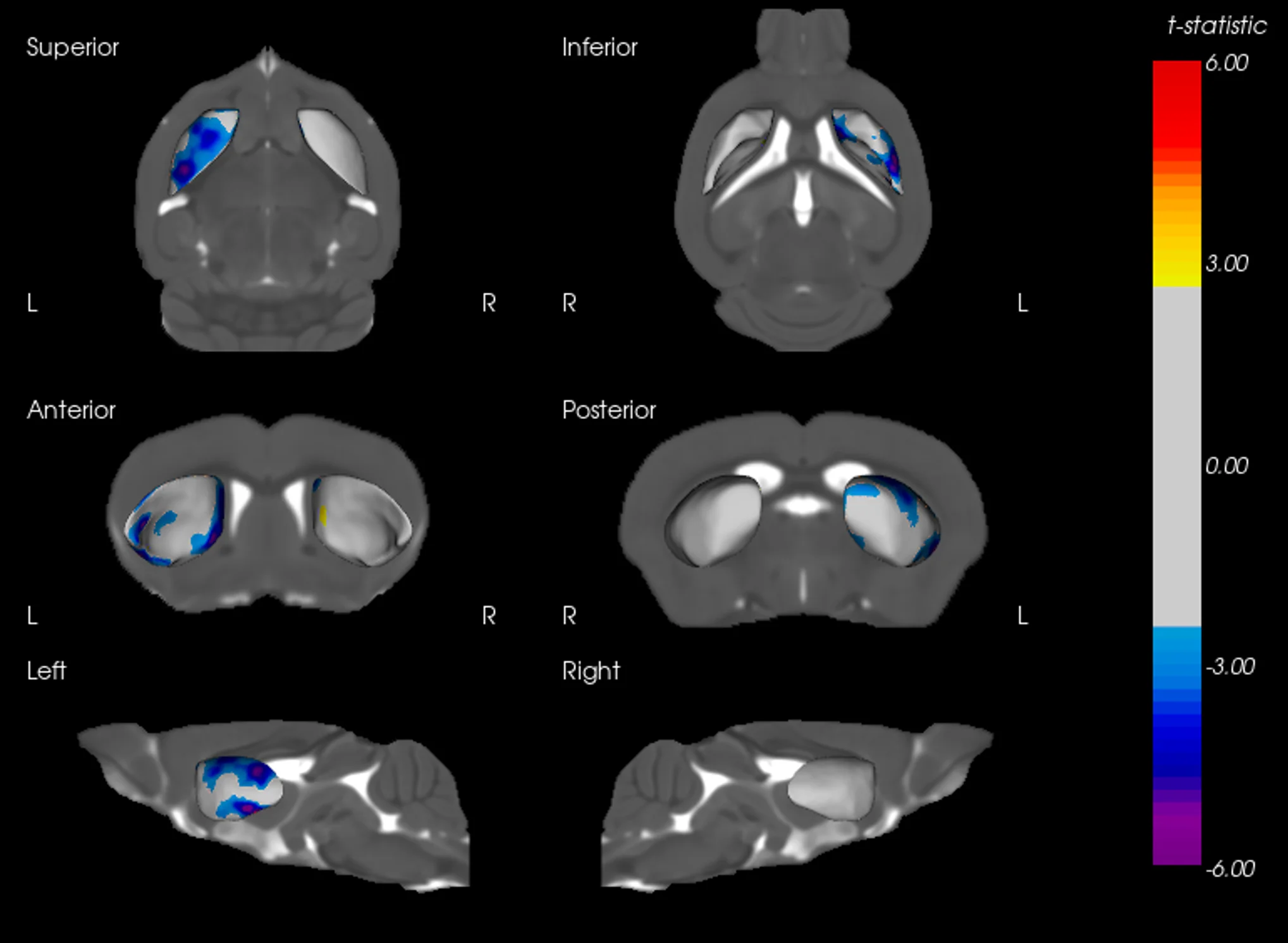
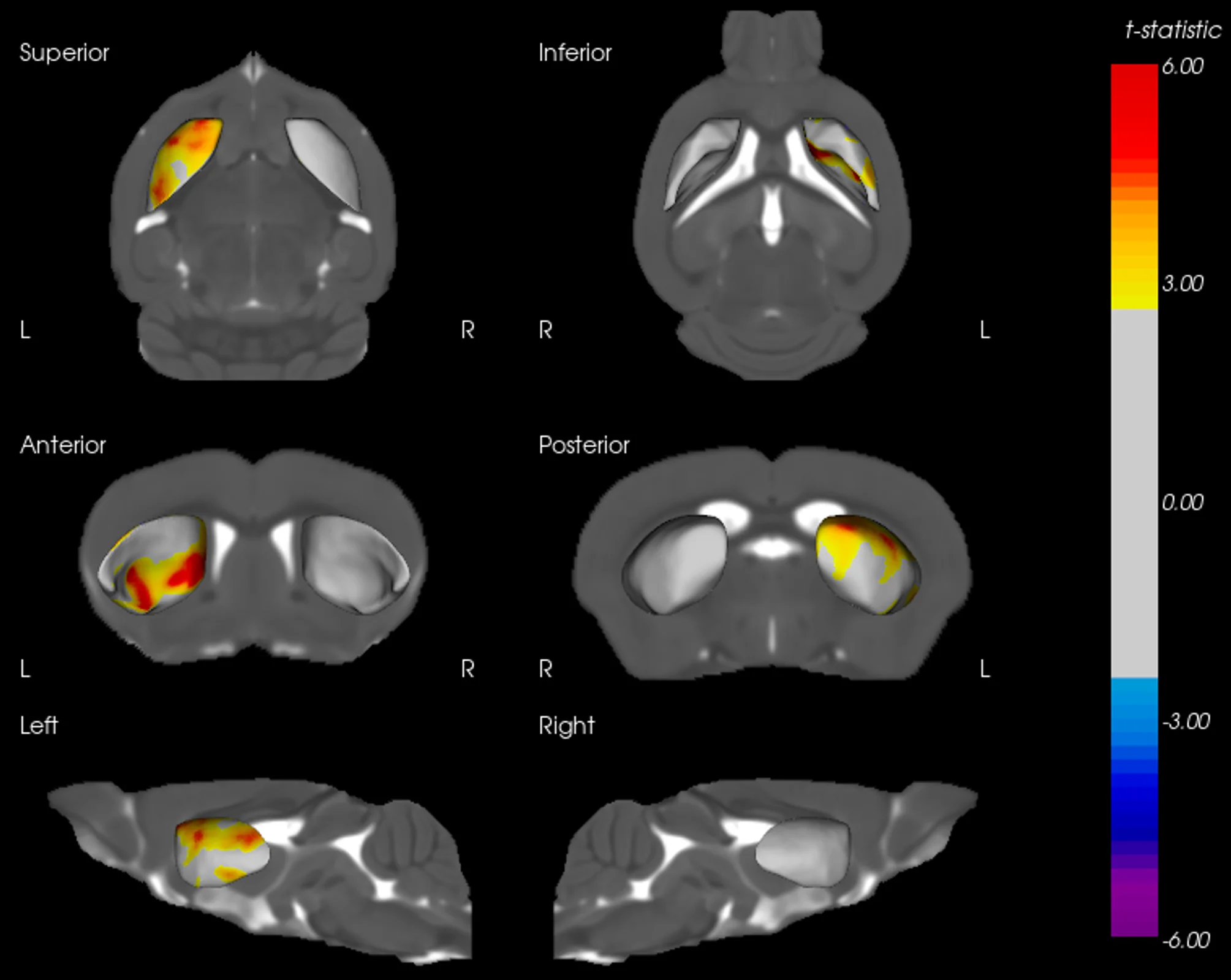
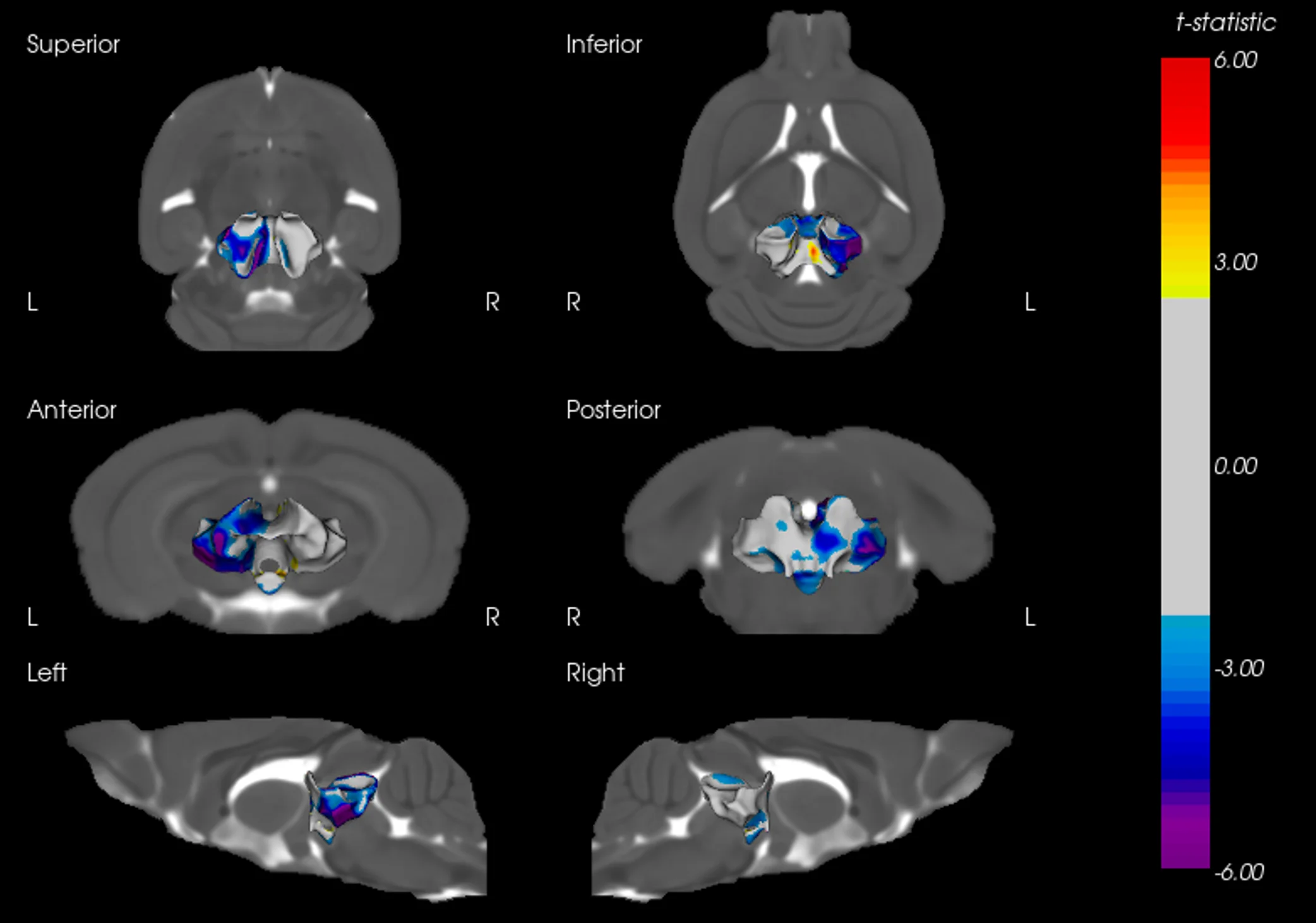
MRI Brain Atrophy: In vivo magnetic resonance imaging to quantify regional brain volume loss (neurodegeneration) over time. Progressive MRI-detected atrophy in the midbrain and connected structures serves as a translational endpoint paralleling human PD. MRI brain atrophy in the midbrain and striatum are observed in human PSP. MRI biomarkers are also powerful for CBD, making this endpoint highly relevant.
-
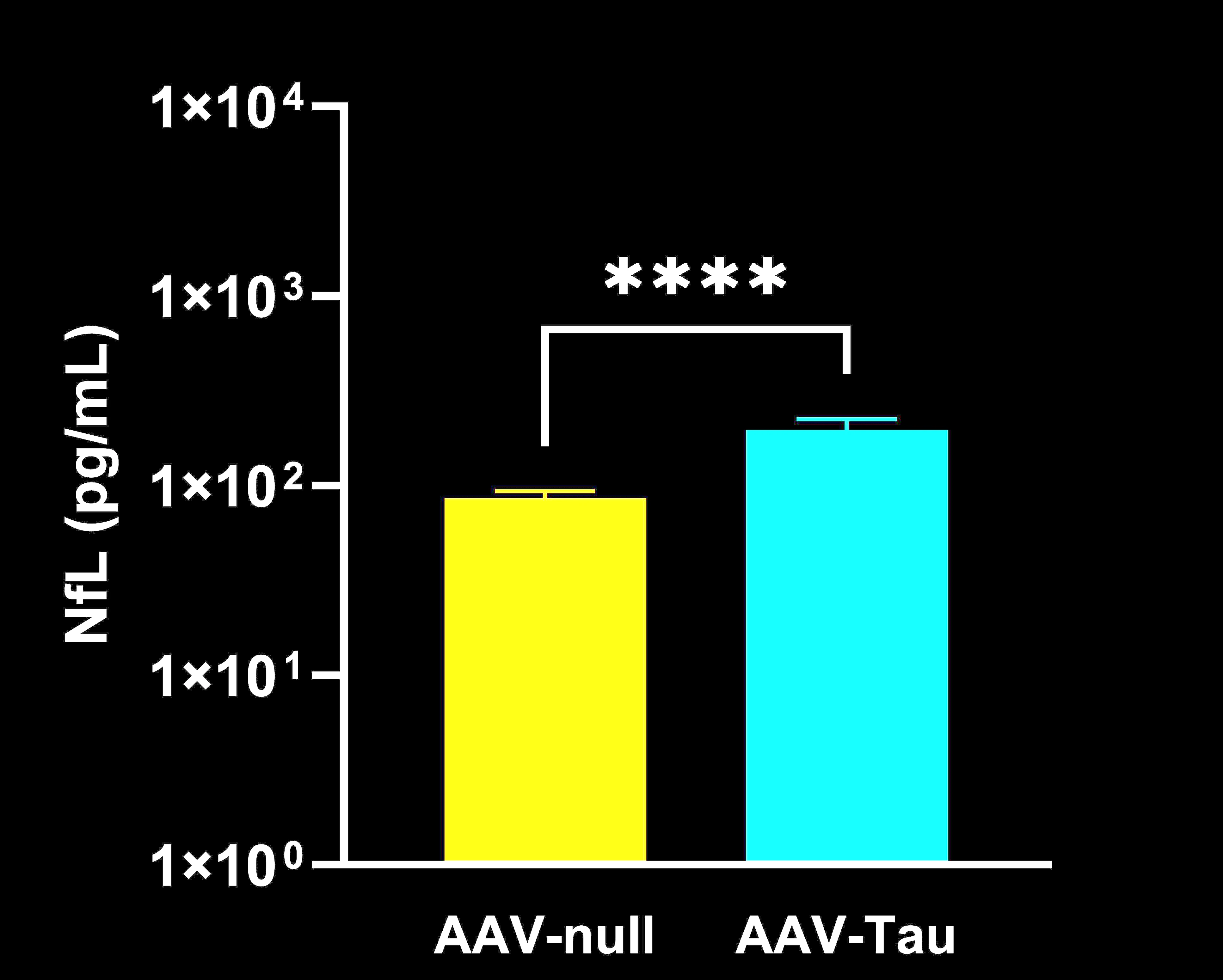
CSF Neurofilament Light Chain (NfL): A fluid biomarker of axonal damage and neurodegeneration, measured in cerebrospinal fluid (and optionally blood plasma). Elevated NfL levels indicate ongoing neuronal injury; this biomarker is also used in clinical trials, making it a valuable bridge between preclinical and clinical results.
-
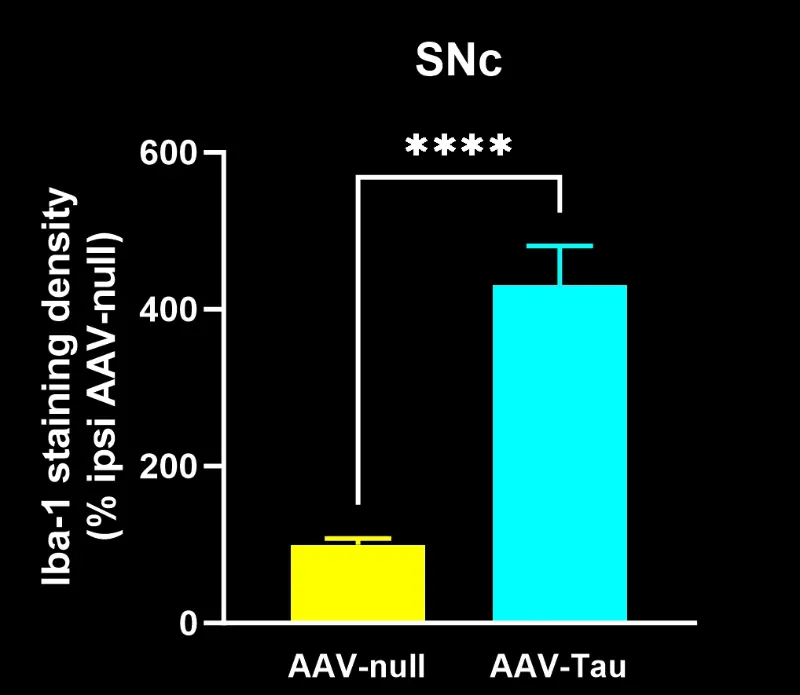
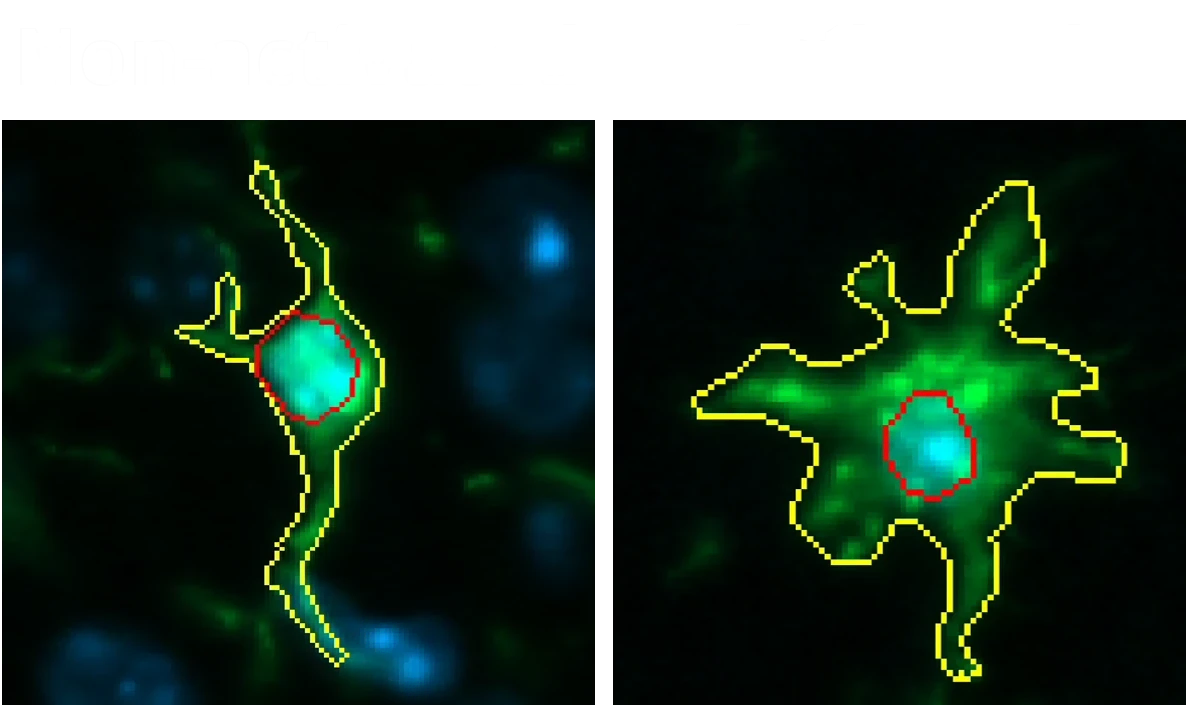
Quantitative Histopathology (IHC/mIF): High-resolution tissue analyses to quantify PD-related pathology. We perform immunohistochemistry (IHC) and multiplex immunofluorescence for markers such as phosphorylated tau inclusions (e.g. AT8), dopaminergic neurons (tyrosine hydroxylase, TH), activated microglia (Iba1), and astrocytes (GFAP). Digital image analysis of these stained tissues provides quantitative measures of neurofibrillary tangle-like aggregates, neuronal loss, and neuroinflammation in the brain.
These endpoints span multiple domains – behavioral, imaging, biochemical, and histological – providing complementary measures of disease severity and therapeutic impact. Notably, the inclusion of translational biomarkers like MRI volumetry and NfL helps bridge preclinical findings to the clinic. By tracking such biomarkers longitudinally in vivo, we can quantitatively monitor disease progression and detect therapeutic effects in a way that is directly relatable to patient outcomes.
In addition to these outcome measures, Biospective distinguishes itself by offering seamless end-to-end integration of all study components. We handle every aspect of the experiment – from viral vector administration, longitudinal behavioral testing, and in vivo MRI/PET imaging to biofluid collection and post-mortem tissue analysis. Our scientific team employs advanced analytics (including automated image analysis for dopaminergic terminal density and AI-driven cell morphology classification) to extract rich datasets from the model. All data are rigorously analyzed and integrated into an interpretable report, allowing you to make informed decisions on your therapeutic candidate’s performance.
Interactive Microscopy Images
Use the Image Viewer below to navigate through high-resolution microscopy images via the left-hand panel or the on-screen arrows. You can pan around the images with your mouse, and zoom in/out using the scroll wheel or the +/- controls. The Control Panel (top-right) allows toggling of image channels and segmentation overlays. For the best experience, we recommend switching to full-screen mode.
This Image Interactive Presentation allows you to explore our characterization of a Novel AAV-hTau mouse model of Tauopathies with Parkinsonian features.
Click to copy link
Biospective's Tau Mouse Model Expertise and Services
Biospective is a global neuroscience CRO with deep expertise in tau animal models – including our proprietary AAV-hTau model, which is a core part of our service portfolio.
Some key advantages of partnering with Biospective for tau model studies include:
-
Extensive Experience & Characterization: We have performed a detailed characterization of the AAV-A53T mouse model through numerous studies, generating datasets that inform best practices and enhance reproducibility. This track record underscores our unique expertise with this tauopathy model.
-
Optimized AAV Vectors & Rapid Study Start: We utilize high-titer, validated AAV vectors encoding human tau to ensure robust, consistent model induction. Biospective maintains ready access to these viral vectors in-house, enabling fast study start-up without delays. Precise stereotaxic injection techniques and optimized dosing result in reliable pathology, and our on-demand vector supply accelerates project timelines.
-
End-to-End Preclinical Services: Biospective provides fully integrated services from initial study design through execution and data analysis. Our capabilities cover all aspects of the project, including surgical model induction (skilled unilateral AAV injections into the substantia nigra), comprehensive in-life assessments (behavioral testing, motor function assays, etc.), in vivo neuroimaging (MRI, PET) for longitudinal monitoring, biofluid collection (CSF, blood) for biomarker analysis, and post-mortem histopathology (immunohistochemistry and multiplex immunofluorescence). This one-stop approach ensures consistency, quality control, and efficient timelines.
- Translational Biomarkers & Readouts: We incorporate clinically relevant biomarkers that bridge preclinical findings to clinical outcomes. For example, we measure neurofilament light chain (NfL) levels in CSF as a biomarker of neurodegeneration (analogous to patient studies), and we perform MRI brain imaging to quantify neurodegenerative atrophy. We also conduct quantitative IHC (e.g. p-Tau, TH for dopaminergic neurons, Iba1 for microglia) and multiplex immunofluorescence to assess pathology and neuroinflammation in tissue. These advanced readouts enhance the translatability of study results to human trials.
-
Global Collaboration & Flexibility: As a global preclinical CRO, we serve biotech and pharmaceutical clients worldwide and tailor each AAV-hTau study to your therapeutic strategy. Our scientists collaborate closely with your team to customize protocols – from adjusting injection parameters (e.g. targeting specific brain regions, unilateral vs. bilateral injections) to incorporating novel endpoints or treatment paradigms. We offer flexibility to meet program-specific needs while maintaining scientific rigor, reproducibility, and transparent communication throughout the partnership.
By leveraging these strengths, Biospective empowers your team to efficiently generate decision-quality data in the AAV hTau model. We pride ourselves on fast project initiation, meticulous data analysis, and supporting our clients through all preclinical phases of tauopathy therapy development.
Contact us to discuss how our Tau mouse models and end-to-end preclinical services can support your Tau drug development program.
Discover more of our Tauopathy Models
Related Content
Up-to-date information on Tauopathies and best practices related to the use of translational biomarkers for the evaluation of therapeutic agents in tau animal models.
Microglia, Astrocytes & Tau in Neurodegenerative Diseases
How glial-driven neuroinflammation fuels tau aggregation, propagation, and neuronal loss in Alzheimer’s disease and other tauopathies.
PS19 Mouse Model for Tau Targeted Drug Development
An overview of PS19 (P301S) mice as a transgenic model for preclinical evaluation of disease-modifying therapies targeting tau seeding & spreading.
A Guide to Mouse Models of Frontotemporal Dementia (FTD)
A Resource providing a comprehensive overview of animal models used in frontotemporal dementia research, including genetic and pathological disease mechanisms.
Tau-related Atrophy is Independent of β-Amyloid & APOE ε4
Using MRI, Tau PET, and Amyloid PET imaging biomarkers from the ADNI study, we show that Tau is more strongly linked to brain atrophy than β-amyloid or APOE ε4.
MRI Measures of Disease Progression for PSP Clinical Trials
MRI biomarkers (including brain atrophy) from the 4RTNI and FTLDNI natural history studies of Progressive Supranuclear Palsy (PSP).
MRI & Corticobasal Degeneration (CBD)
Longitudinal MRI brain atrophy measures from the 4RTNI and FTLDNI studies including sample size calculations for clinical trials of Corticobasal Degeneration.
Decreased Brain Glucose Metabolism in MCI is Driven by Tau
We demonstrate a more significant role of tau than β-amyloid in the reduction of cerebral glucose metabolism by analysis of PET images from the ADNI study.
Autophagy and Transcription Factor EB (TFEB)
An overview of Transcription Factor EB (TFEB) and its role in autophagy and neurodegenerative diseases.
Autophagy & Neurodegenerative Diseases
An overview of how cellular autophagy plays a role in brain health and neurodegeneration.
Inflammasome – A Therapeutic Target for Multiple Diseases
An overview of inflammasomes, including their mechanisms of action, roles in diseases, and targeting for drug development.
What Is IL-1β (IL-1b)? Function, Signaling, and Biological Role
An overview of IL-1β, including its signaling pathways, involvement in disease mechanisms, and potential therapeutic targets.
What is NF-κB (Nuclear Factor Kappa B)?
An overview of NF-κB, highlighting its role in inflammation and diseases (including neurological disorders), and therapeutic strategies targeting NF-κB.
What is NLRP3?
An overview of NLRP3 inflammasome activation triggers, disease associations, and therapeutic targeting strategies.
Impaired Microglia Autophagy in Neurodegenerative Diseases
How impaired microglia autophagy contributes to the progression of neurodegenerative diseases.
TNF-α (TNF-alpha) & Microglia in Neurodegenerative Diseases
An overview of the function of tumor necrosis factor-alpha (TNF-α) in microglia and its contribution to the progression of neurodegeneration.
NLRP3 Inflammasome and Neurodegenerative Diseases
An overview of the NLRP3 inflammasome and its role in neurodegenerative diseases, including Alzheimer's disease, Parkinson’s disease, and ALS.
Lysosome Dysfunction in Microglia & Astrocytes
An overview of lysosomal dysfunction in microglia & astrocytes, and its role in neurodegenerative diseases.
Interleukin-1 Beta (IL-1β) and Neurodegenerative Diseases
The role of IL-1beta in neurodegenerative diseases, including Alzheimer's disease (AD), Parkinson’s disease (PD), and amyotrophic lateral sclerosis (ALS).