Synuclein Model Overview
Alpha-synuclein (α-synuclein; α-syn) is a major pathogenic protein found in Lewy bodies and neurites in the substantia nigra pars compacta and other brain regions in Parkinson's disease (PD). For this Parkinson's disease animal model, we perform stereotaxic inoculation of recombinant, human α-syn preformed fibrils (PFFs) into brains of M83 transgenic mice [B6;C3-Tg( Prnp SNCA A53T )83Vle/J] which overexpress α-synuclein, or we inject recombinant murine α-syn into the brains of non-transgenic, wild-type (B6-C3H) mice.
Our validated injection sites are: Anterior Olfactory Nucleus (AON), Striatum +/- Overlying Cerebral Cortex, and Medial Forebrain Bundle (MFB).
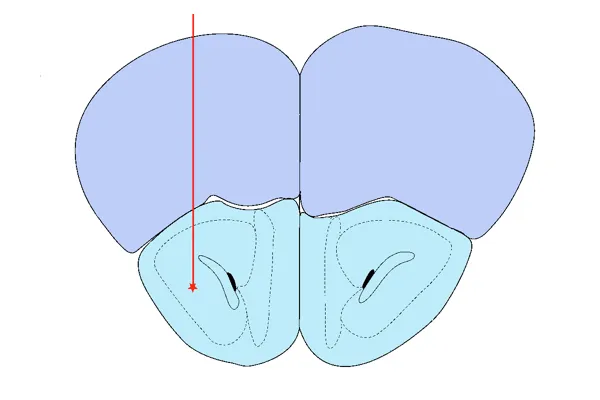
AON Injection
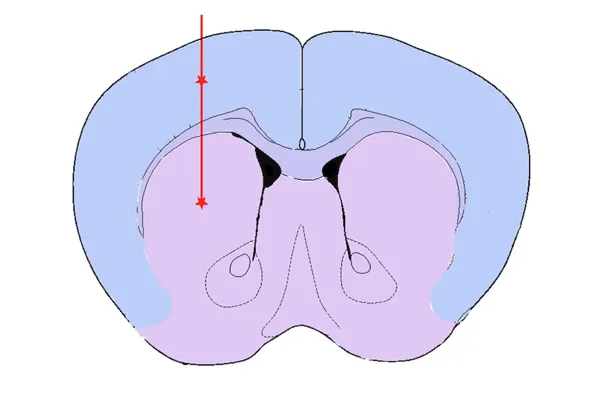
Striatum & Overlying Cortex Injection
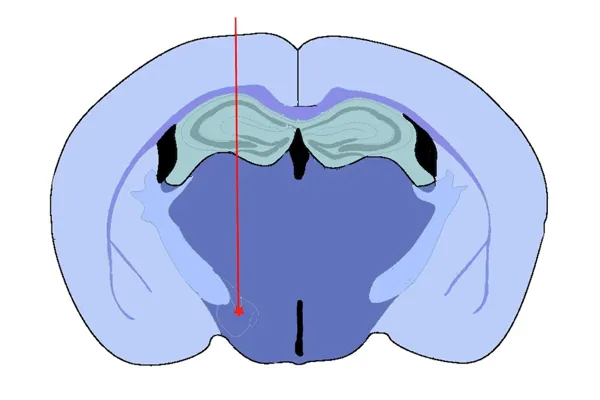
Medial Forebrain Bundle (MFB) Injection
Spatial pattern of spread of α-synuclein pathology following stereotaxic injection of recombinant preformed fibrils into the anterior olfactory nucleus (AON).
The rationale for targeting the Anterior Olfactory Nucleus (AON) includes:
- It is one of the earliest sites to demonstrate α-synuclein pathology in human Parkinson's disease (Braak Stage 1)
- Non-motor symptoms associated with the limbic system (e.g. hyposmia/anosmia, sleep alterations) are prevalent, early symptoms of Parkinson's disease
- The AON is connected to several ipsilateral & contralateral brain regions, including direct (1st order) connections to olfactory bulb, amygdala, piriform cortex, and higher order connections, such as the entorhinal cortex and hippocampus; many of these regions are highly vulnerable to neurodegeneration
The rationale for targeting the Striatum +/- Overlying Cortex includes:
- It is the injection site originally reported (Luk et al., J. Exp. Med., 209: 975-986, 2011; doi: 10.1084/jem.20112457; Luk et al., Science, 338: 949-953, 2012; doi: 10.1126/science.1227157)
- The dopaminergic nerve terminals in the striatum originate from neurons in the substantia nigra
- Injection into the overlying cerebral cortex increases the extent alpha-synuclein pathology in the brain
Our rationale for targeting the Medial Forebrain Bundle (MFB) includes:
- It is a tract directly connected to the substantia nigra and ventral tegmental area, and is often targeted in toxin-based models of human Parkinson's disease (e.g. 6-hydroxydopamine [6-OHDA])
- It serves as a "hub" for projections from many other brain regions, thereby serving as an excellent seeding location to facilitate spreading of alpha-synuclein pathology to distant brain regions
- This model results in loss of tyrosine hydroxylase-positive dopaminergic neurons in the substantia nigra and associated motor deficits
A significant advantage of this animal model of Parkinson's disease is that it reproduces many aspects of the human disease, including:
- Highly reproducible spreading of alpha-synuclein in a well-defined spatiotemporal pattern, resulting in extensive alpha-synuclein pathology in neuronal cell bodies and neurites
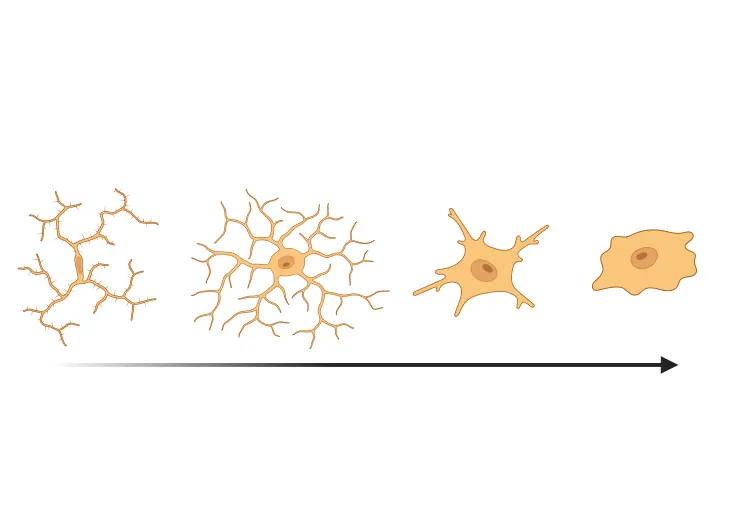
- Strong neuroinflammation (activated microglia and reactive astrocytes)
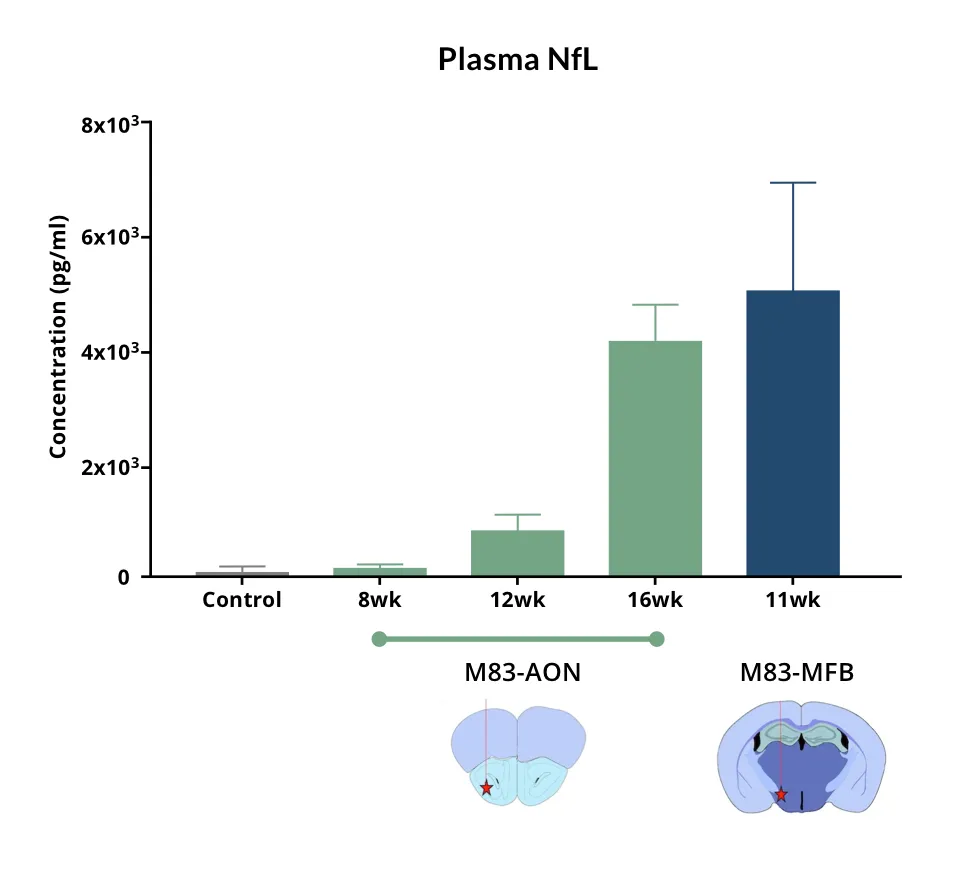
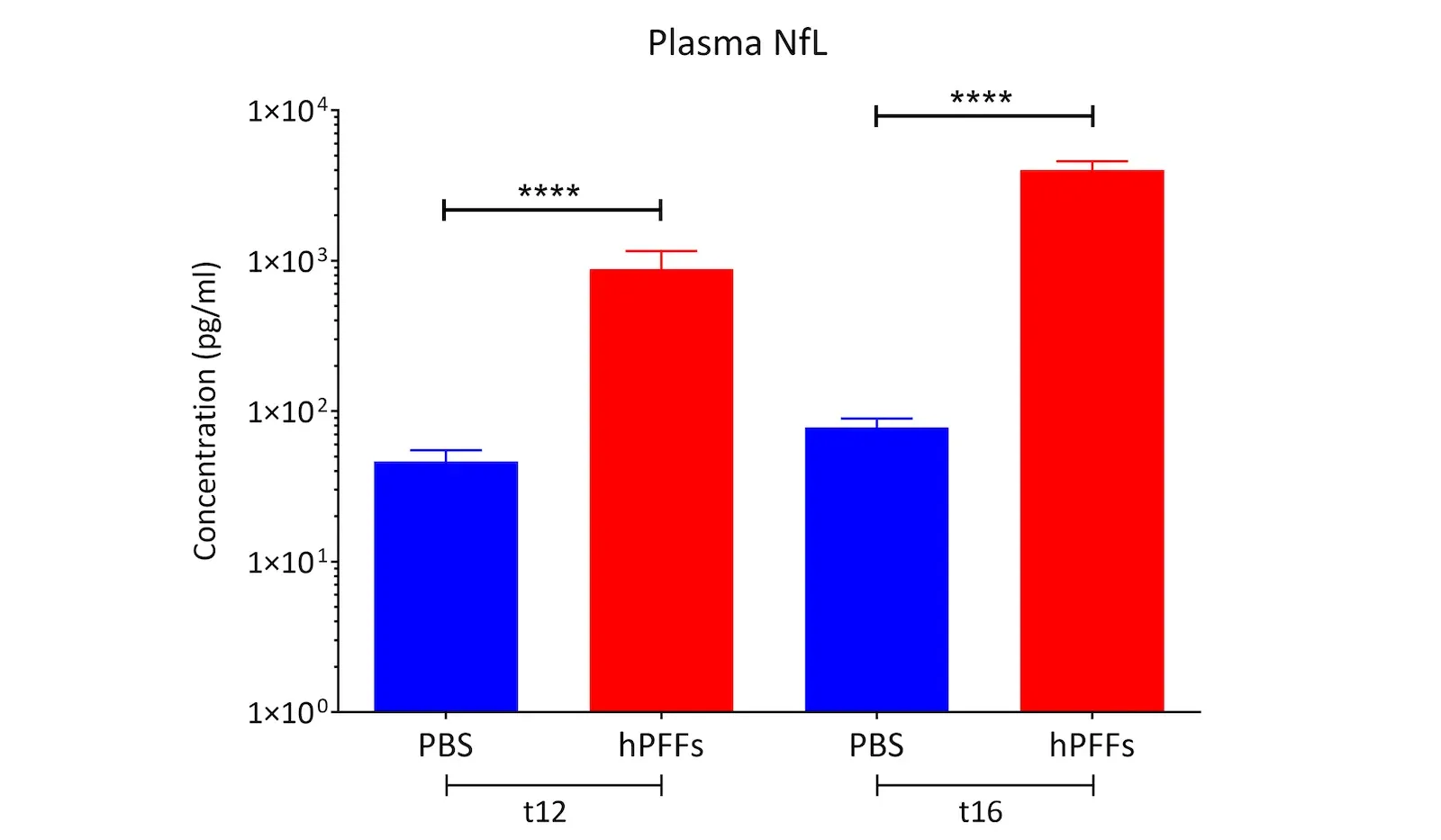
- Neurodegeneration with increased neurofilament light chain (NfL; NF-L) levels in the blood & CSF
- Measurable behavioral changes (e.g. motor function, sleep)
- Amenable to disease modification via therapeutic intervention
Synuclein Model Generation
A general schema for synuclein PFF animal model generation is:
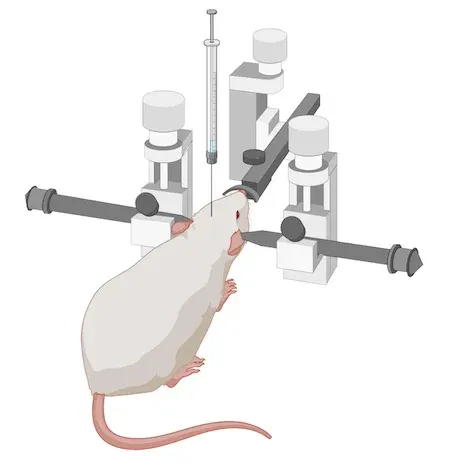
We have in-licensed the M83 transgenic (tg) mice. As a Preclinical Neuroscience CRO, we maintain a well-established breeding colony of these transgenic alpha-synuclein mice in-house at Biospective, allowing us to conduct large-scale (>100 mice) studies.
For this rodent model, we breed and age M83+/- mice to 8-12 weeks-of-age. Alternatively, we use wild-type (B6-C3H) mice at 8-12 weeks-of-age. We then perform stereotaxic injection of sonicated, recombinant human alpha-synuclein PFFs into the target region. We use digital stereotaxic devices with automated microinjectors for high accuracy & precision.
This animal model is highly reproducible with a nearly 100% success rate of synuclein PFF seeding.
Our Synuclein Mouse Model Validated Measures
- Body weight
- Clinical scores
- Behavioral assessments
- Motor function tests
- Sleep analysis
- MRI brain atrophy (Read More in our Presentation - Brain Atrophy Analysis in Mouse Models of Neurodegeneration)
- Neurofilament light chain measures in plasma & CSF (Read More in our Resource - Neurofilament Light in Parkinson's Disease Models)
- Immunohistochemistry & multiplex immunofluorescence (Read More in our Presentation - Microglial Activation in an α-Synuclein Mouse Model of Parkinson's Disease)
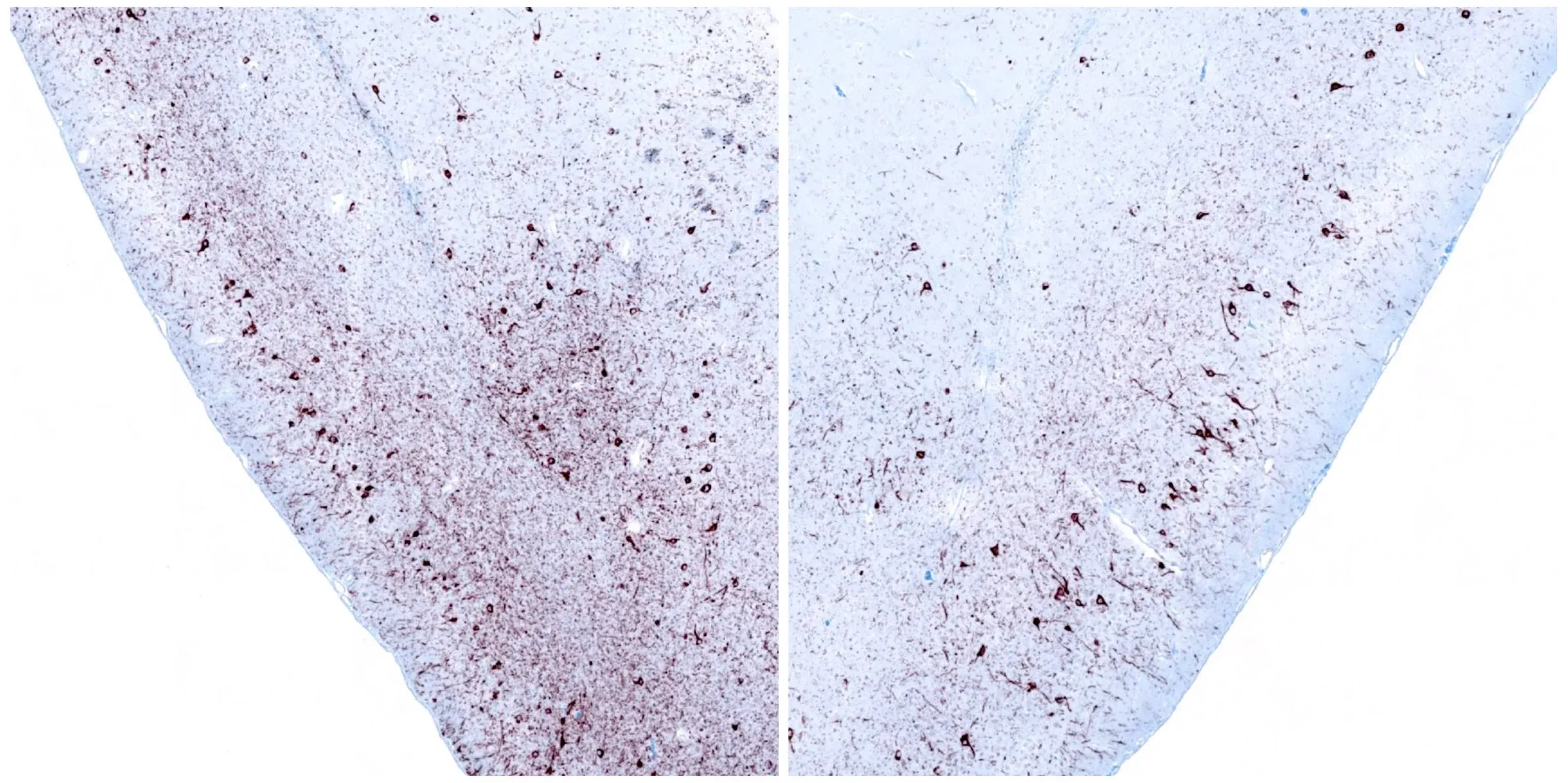
Phosphorylated α-synuclein immunohistochemistry staining of the ipsilateral (left) and contralateral (right) piriform cortex at 12 weeks following unilateral stereotaxic injection of α-synuclein preformed fibrils into the anterior olfactory nucleus of an M83+/- mouse.
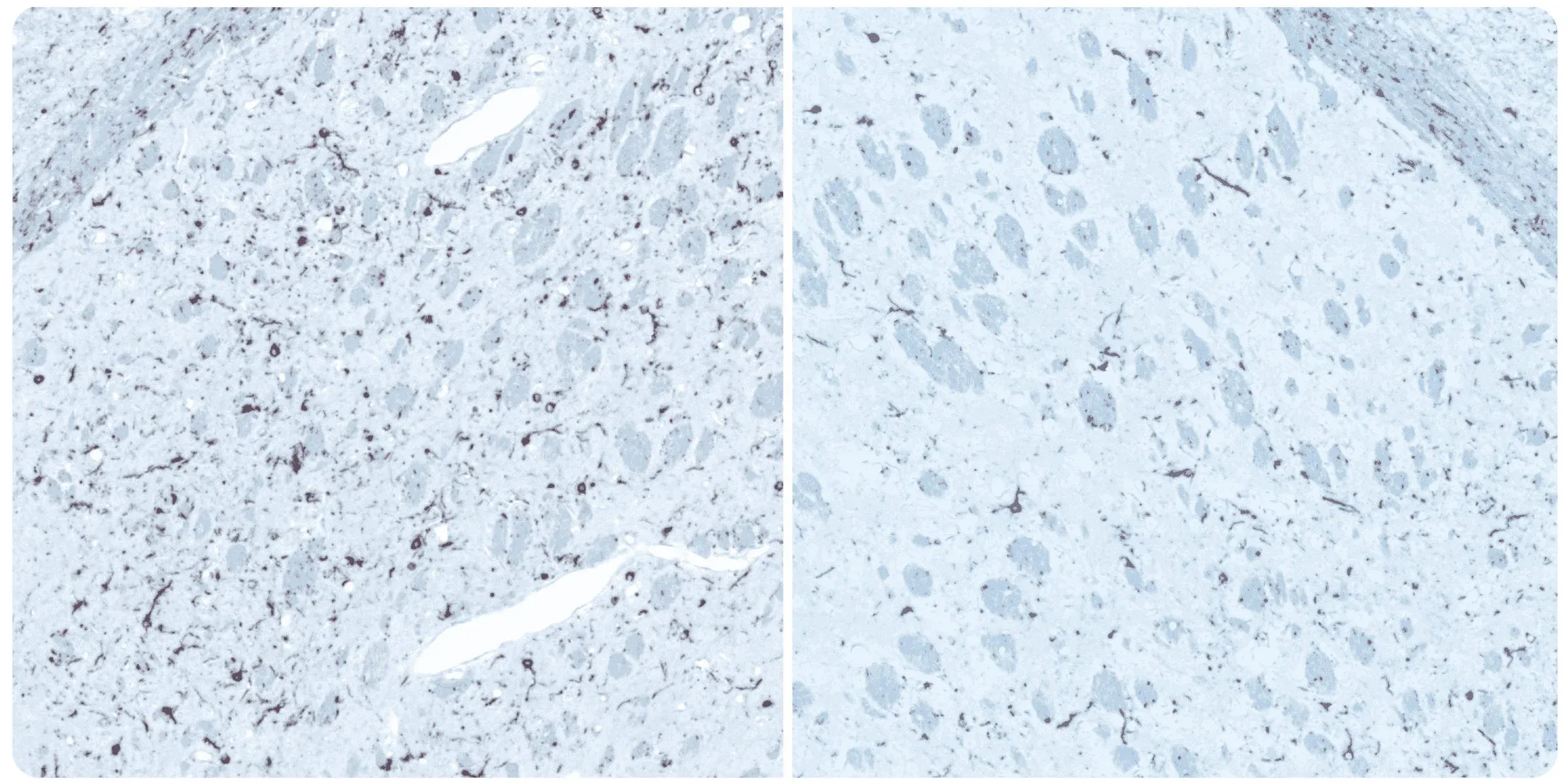
Phosphorylated alpha-synuclein immunohistochemistry staining of the ipsilateral (left) and contralateral (right) striatum at 12 weeks post-injection following unilateral stereotaxic inoculation of α-synuclein preformed fibrils (PFFs) into the striatum and overlying cerebral cortex of an M83+/- mouse.
Learn more about our characterization of this model, our validated measures, and our Preclinical Neuroscience CRO services.
Discover more of our Parkinson's Disease Models
Related Content
Up-to-date information on Parkinson's Disease and best practices related to the evaluation of therapeutic agents in PD animal models.
Neurofilament Light Chain in Parkinson's Disease Models
How neurofilament light chain (NfL; NF-L) levels can be used as blood (plasma; serum) & CSF biomarkers in Parkinson's disease mouse and rat models.
AAV α-Synuclein Models for Parkinson’s Disease Drug Development
Overview of adeno-associated virus (AAV) induced α-synuclein expression in mouse & rat models for use in preclinical studies of disease-modifying therapeutics.
Microglial Activation in an α-Synuclein PFF Mouse Model
We have quantified microglial activation, based on morphology, in an α-synuclein preformed fibril (PFF) seeding & spreading mouse model of Parkinson’s disease.